D5314
DPQ
≥98% (HPLC), solid
Sinónimos:
3,4-Dihydro-5-[4-(1-piperidinyl)butoxyl]-1(2H)-isoquinolinone
About This Item
Productos recomendados
biological source
synthetic (organic)
Quality Level
assay
≥98% (HPLC)
form
solid
mp
107-109 °C
solubility
DMSO: 1 mg/mL, clear, colorless to faintly yellow
storage temp.
2-8°C
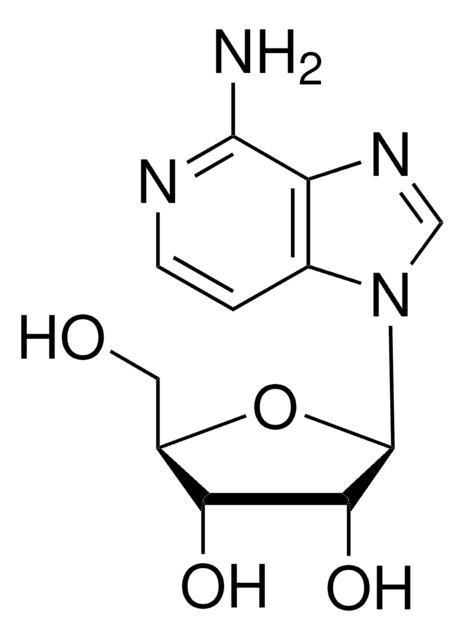
SMILES string
N3(CCCCC3)CCCCOc1c2c(ccc1)C(=O)NCC2
InChI
1S/C18H26N2O2/c21-18-16-7-6-8-17(15(16)9-10-19-18)22-14-5-4-13-20-11-2-1-3-12-20/h6-8H,1-5,9-14H2,(H,19,21)
InChI key
RVOUDNBEIXGHJY-UHFFFAOYSA-N
Application
Biochem/physiol Actions
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico