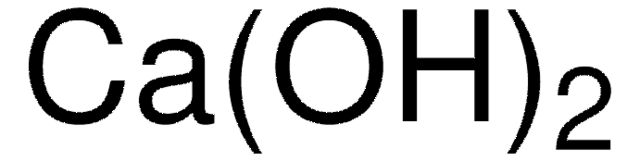239232
Calcium hydroxide
ACS reagent, ≥95.0%
About This Item
Productos recomendados
grade
ACS reagent
assay
≥95.0%
form
powder
impurities
≤0.03% insol. in HCl
≤0.1% S compounds
≤3.0% calcium carbonate
pH
12.4-12.6 (20 °C)
density
2.24 g/mL at 25 °C (lit.)
anion traces
chloride (Cl-): ≤0.03%
cation traces
Fe: ≤0.05%
K: ≤0.05%
Mg: ≤0.5%
Na: ≤0.05%
Sr: ≤0.05%
heavy metals: ≤0.003% (by ICP)
SMILES string
O[Ca]O
InChI
1S/Ca.2H2O/h;2*1H2/q+2;;/p-2
InChI key
AXCZMVOFGPJBDE-UHFFFAOYSA-L
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
- In the pretreatment of technical lignin during the pyrolysis in a fluidized bed reactor to overcome the agglomeration to produce phenolic-rich bio-oil.
- As a catalyst in the pyrolysis of pinewood in a fixed bed reactor.
signalword
Danger
hcodes
Hazard Classifications
Eye Dam. 1 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
13 - Non Combustible Solids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico