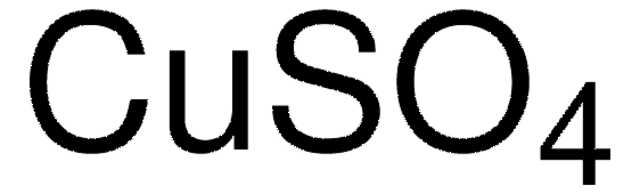209198
Copper(II) sulfate pentahydrate
ACS reagent, ≥98.0%
Sinónimos:
Cupric sulfate pentahydrate, Sulfuric acid, copper(II) salt (1:1) pentahydrate
About This Item
Productos recomendados
grade
ACS reagent
Quality Level
vapor pressure
7.3 mmHg ( 25 °C)
assay
≥98.0%
98.0-102.0% (ACS specification)
form
crystals
reaction suitability
reaction type: click chemistry
reagent type: catalyst
core: copper
pH
3.5-4.5 (20 °C, 50 g/L)
mp
110 °C (dec.) (lit.)
anion traces
chloride (Cl-): ≤0.001%
cation traces
Ca: ≤0.005%
Fe: ≤0.003%
K: ≤0.01%
Na: ≤0.02%
Ni: ≤0.005%
SMILES string
O.O.O.O.O.[Cu++].[O-]S([O-])(=O)=O
InChI
1S/Cu.H2O4S.5H2O/c;1-5(2,3)4;;;;;/h;(H2,1,2,3,4);5*1H2/q+2;;;;;;/p-2
InChI key
JZCCFEFSEZPSOG-UHFFFAOYSA-L
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
- In acetylation of alcohols and phenols in the presence of acetic anhydride.
- To synthesize various ynamides via intramolecular coupling of amides with alkynyl bromides.
It can also be used as a template in the synthesis of copper(II) ion-imprinted polymers using vinylpyridine and methacrylic acid as functional monomers. This polymer is used to quantify Cu2+ ion by using flame atomic absorption spectroscopy.
Features and Benefits
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Acute 1 - Aquatic Chronic 1 - Eye Dam. 1
Storage Class
13 - Non Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico