94876
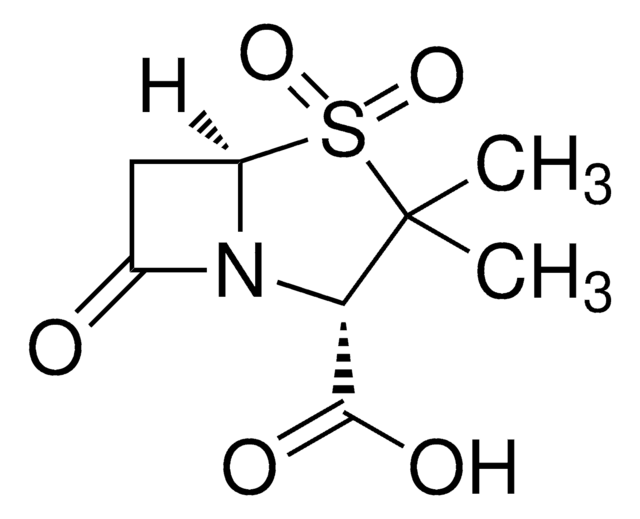
Sulbactam
analytical standard
Sinónimos:
(2S,5R)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid 4,4-dioxide, Betamaze, CP 45899, CP-45,899, Penicillanic acid 1,1-dioxide, Penicillanic acid S,S-dioxide, Penicillanic acid dioxide, Penicillanic acid sulfone
About This Item
Productos recomendados
grade
analytical standard
Quality Level
assay
≥99.0% (HPLC)
shelf life
limited shelf life, expiry date on the label
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
application(s)
forensics and toxicology
pharmaceutical (small molecule)
format
neat
storage temp.
2-8°C
SMILES string
CC1(C)[C@@H](N2[C@@H](CC2=O)S1(=O)=O)C(O)=O
InChI
1S/C8H11NO5S/c1-8(2)6(7(11)12)9-4(10)3-5(9)15(8,13)14/h5-6H,3H2,1-2H3,(H,11,12)/t5-,6+/m1/s1
InChI key
FKENQMMABCRJMK-RITPCOANSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico