31581
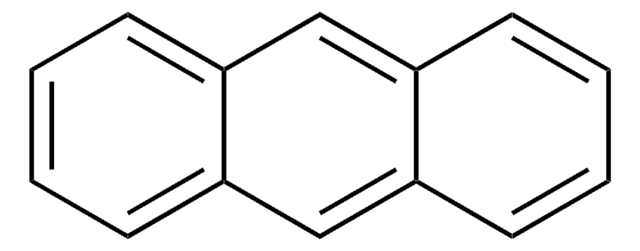
Anthracene
analytical standard
Sinónimos:
Anthraxcene, Paranaphthalene
About This Item
Productos recomendados
grade
analytical standard
Quality Level
vapor density
6.15 (vs air)
vapor pressure
1 mmHg ( 145 °C)
autoignition temp.
1004 °F
shelf life
limited shelf life, expiry date on the label
technique(s)
HPLC: suitable
gas chromatography (GC): suitable
bp
340 °C (lit.)
mp
210-215 °C (lit.)
solubility
alcohols: soluble
benzene: soluble
chloroform: soluble
hydronaphthalenes: soluble
supercritical carbon dioxide: soluble
application(s)
environmental
format
neat
SMILES string
c1ccc2cc3ccccc3cc2c1
InChI
1S/C14H10/c1-2-6-12-10-14-8-4-3-7-13(14)9-11(12)5-1/h1-10H
InChI key
MWPLVEDNUUSJAV-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
Recommended products
signalword
Warning
hcodes
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Eye Irrit. 2
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
flash_point_f
249.8 °F - closed cup
flash_point_c
121.0 °C - closed cup
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Protocolos
US EPA Method 8270 (PAH only): GC Analysis of PAHs on SLB®-5ms
US EPA Method 610 describes the analysis of polynuclear aromatic hydrocarbons (commonly referred to as PAHs or PNAs) by both HPLC and GC.
HPLC Analysis of PAHs on SUPELCOSIL™ LC-PAH
GC Analysis of Polynuclear Aromatic Hydrocarbons (PAHs) in Salmon on SPB®-608 (20 m x 0.18 mm I.D., 0.18 µm) after QuEChERS Cleanup using Supel™ QuE Z-Sep, Fast GC Analysis
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico