745979
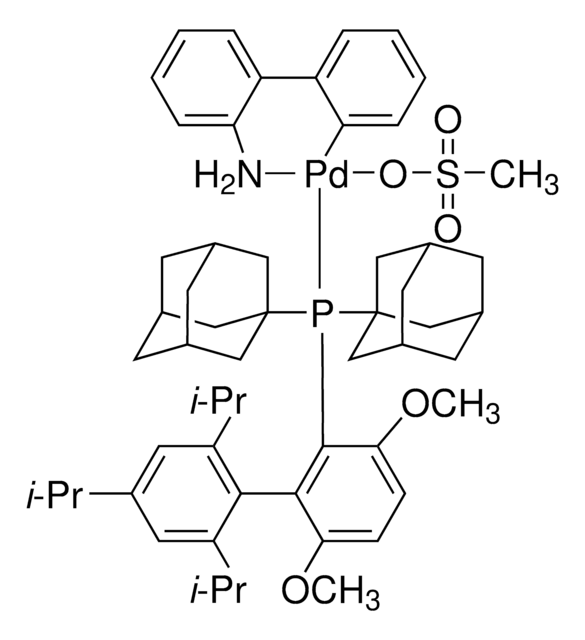
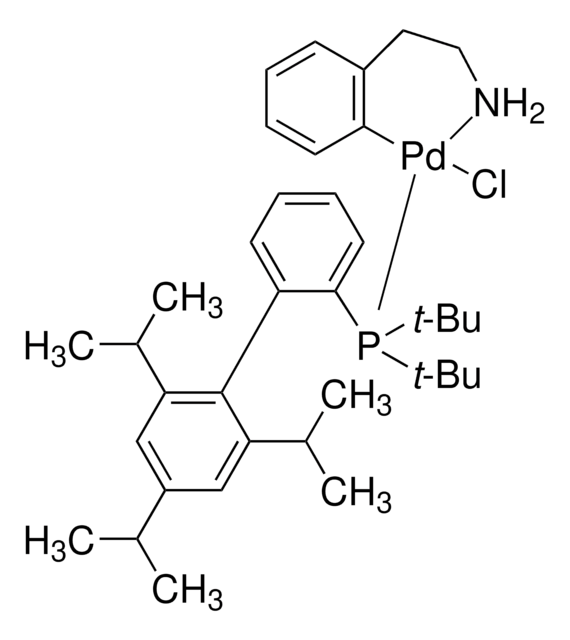
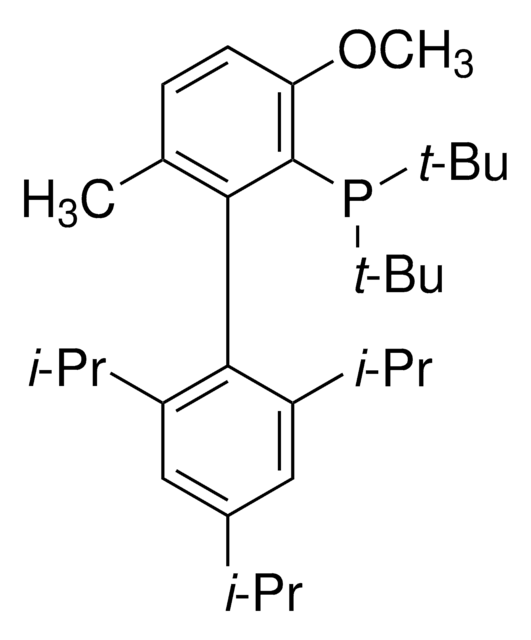
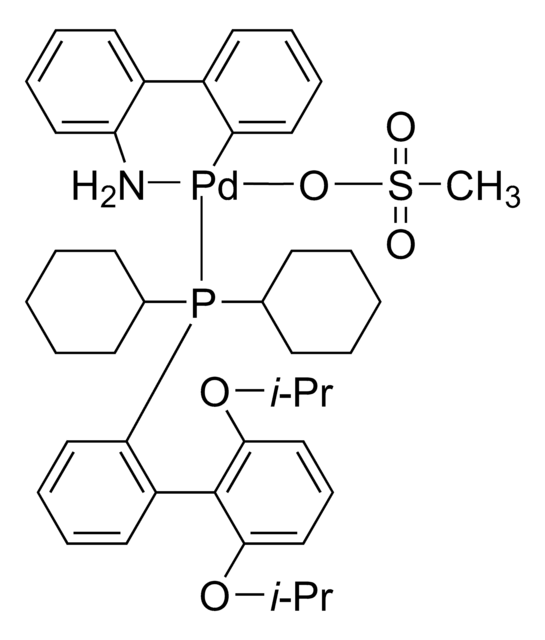
tBuBrettPhos Pd G3
96%
Sinónimos:
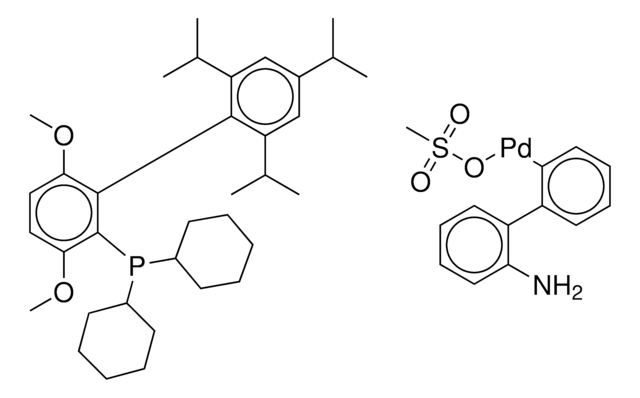
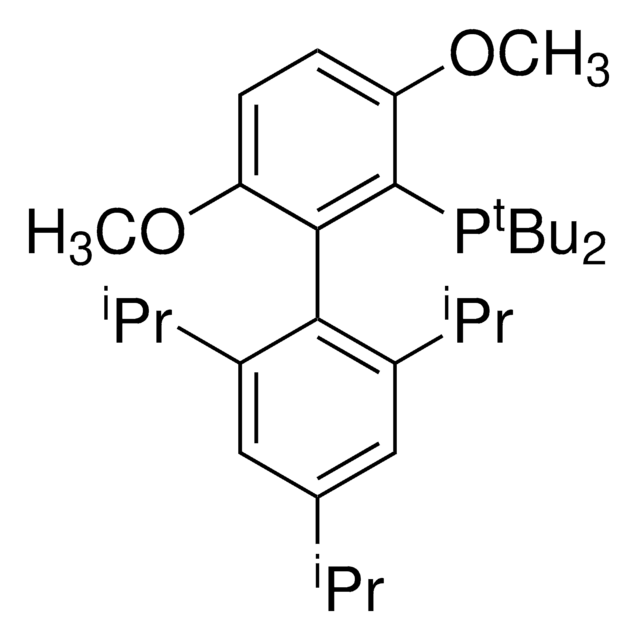
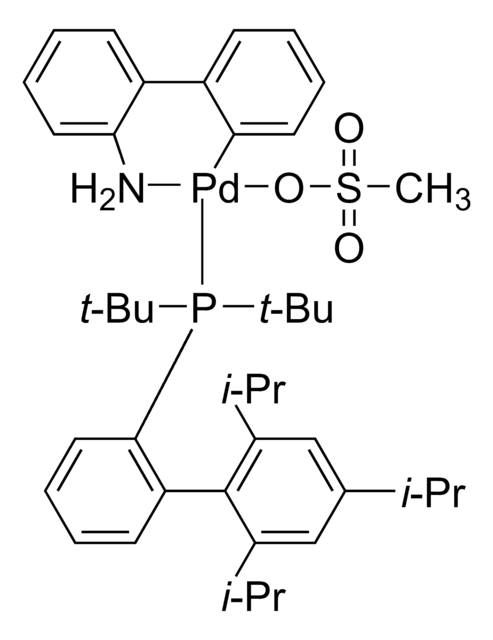
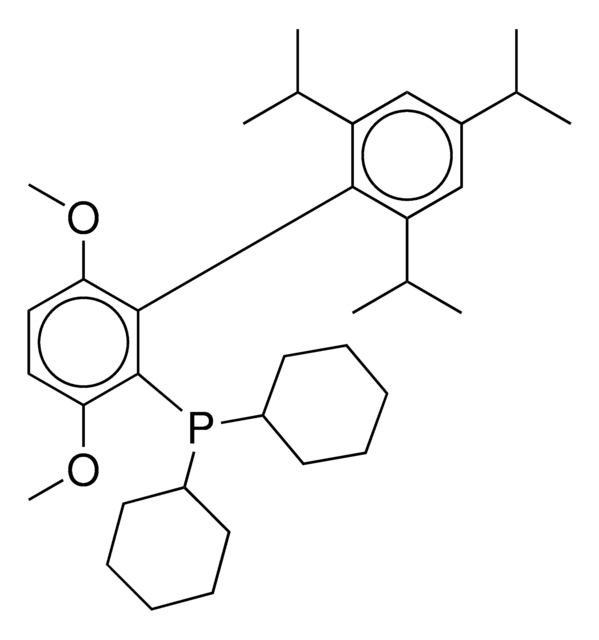
tert-BuBrettPhos-Pd-G3, [(2-Di-tert-butylphosphino-3,6-dimethoxy-2′,4′,6′-triisopropyl-1,1′-biphenyl)-2-(2′-amino-1,1′-biphenyl)]palladium(II) methanesulfonate
About This Item
Productos recomendados
Quality Level
assay
96%
form
solid
feature
generation 3
reaction suitability
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
reaction type: Cross Couplings
mp
119-131 °C
functional group
phosphine
SMILES string
COC1=CC=C(C(P(C(C)(C)C)C(C)(C)C)=C1C2=C(C=C(C=C2C(C)C)C(C)C)C(C)C)OC.NC3=C(C=CC=C3)C4=C(C=CC=C4)[Pd]OS(C)(=O)=O
InChI
1S/C31H49O2P.C12H10N.CH4O3S.Pd/c1-19(2)22-17-23(20(3)4)27(24(18-22)21(5)6)28-25(32-13)15-16-26(33-14)29(28)34(30(7,8)9)31(10,11)12;13-12-9-5-4-8-11(12)10-6-2-1-3-7-10;1-5(2,3)4;/h15-21H,1-14H3;1-6,8-9H,13H2;1H3,(H,2,3,4);/q;;;+1/p-1
InChI key
GAQPAUHHECNHNS-UHFFFAOYSA-M
General description
Application
For small scale and high throughput uses, product is also available as ChemBeads (928313)
Related product
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
G3 and G4 Buchwald palladium precatalysts are the newest air, moisture, and thermally stable crossing-coupling complexes used in bond formation for their versatility and high reactivity.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico