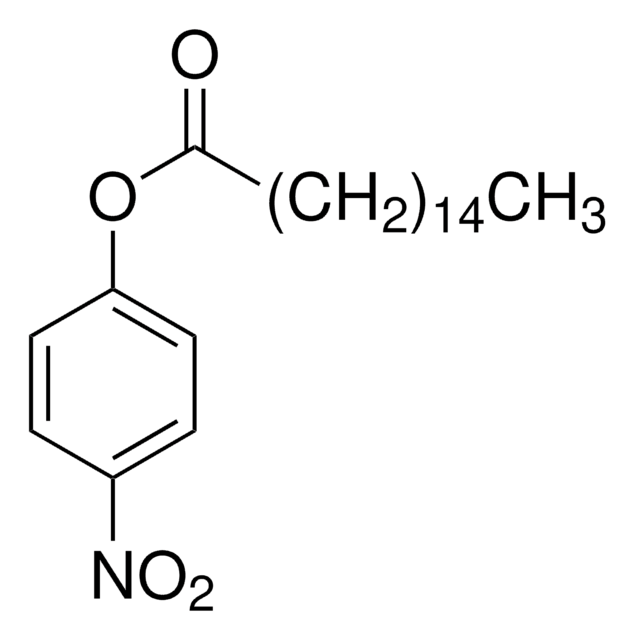
701122
Cloruro de hierro (III)
sublimed grade, ≥99.9% trace metals basis
Sinónimos:
Cloruro férrico, Molisita, Tricloruro de hierro
About This Item
Productos recomendados
grade
sublimed grade
Quality Level
vapor density
5.61 (vs air)
vapor pressure
1 mmHg ( 194 °C)
assay
≥99.9% trace metals basis
form
powder or crystals
reaction suitability
reagent type: catalyst
core: iron
greener alternative product characteristics
Catalysis
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
technique(s)
cell culture | mammalian: suitable
impurities
≤1000.0 ppm Trace Metal Analysis
mp
304 °C (lit.)
application(s)
battery manufacturing
greener alternative category
SMILES string
Cl[Fe](Cl)Cl
InChI
1S/3ClH.Fe/h3*1H;/q;;;+3/p-3
InChI key
RBTARNINKXHZNM-UHFFFAOYSA-K
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
Application
- As a precursor to prepare Fe(III)-chlorophyll complex. Fe(III) enhances the performance of chlorophyll as a dye sensitizer in DSSCs by forming complex compounds with metal-ligand charge transfer properties, leading to increased efficiency and improved paramagnetic properties.
- As a catalyst /modifying agent to prepare high performance porous carbon for lithium-ion battery anodes. The addition of FeCl3 enhances the graphitization of porous carbon without significantly affecting the layer spacing and also the specific surface area and pore volume of porous carbon.
- As a co-catalyst to fabricate liquid catalyzed fuel cell (LCFC) for direct conversion from carbohydrates to electricity. It helps to improve the hydrolysis of carbohydrate and enhances the electron transfer from carbohydrates to anode.
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Eye Dam. 1 - Met. Corr. 1 - Skin Irrit. 2
Storage Class
8B - Non-combustible corrosive hazardous materials
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Faceshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Professor Randal Lee (University of Houston, USA) discusses design considerations for iron oxide magnetic nanospheres and nanocubes used for biosensing, including synthetic procedures, size, and shape. The effects of these variables are discussed for various volumetric-based and surface-based detection schemes.
Lithium-Ion Battery Performance: Dependence on Material Synthesis and Post‑Treatment Methods
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico