658235
Lithium dimethylaminoborohydride solution
1 M in THF
Sinónimos:
N-Methylmethanamine boron complex, Lithium (dimethylamino)trihydroborate, Lithium trihydro(N-methylmethanaminato)borate
About This Item
Productos recomendados
reaction suitability
reagent type: reductant
concentration
1 M in THF
refractive index
n20/D 1.423
density
0.882 g/mL at 25 °C
storage temp.
2-8°C
SMILES string
[Li+].[BH3-]N(C)C
InChI
1S/C2H9BN.Li/c1-4(2)3;/h1-3H3;/q-1;+1
InChI key
CEDUMRZWZLVFKS-UHFFFAOYSA-N
Categorías relacionadas
General description
Application
Reactant for:
- B-H oxidative addition reactions
- Reduction and amination reactions
- Reduction of N-alkyl lactams
- Synthesis of tertiary amine-boranes
signalword
Danger
hcodes
Hazard Classifications
Acute Tox. 4 Oral - Carc. 2 - Eye Irrit. 2 - Flam. Liq. 2 - STOT SE 3
target_organs
Respiratory system
supp_hazards
Storage Class
4.3 - Hazardous materials which set free flammable gases upon contact with water
wgk_germany
WGK 3
flash_point_f
1.4 °F - closed cup
flash_point_c
-17 °C - closed cup
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Artículos
Lithium aminoborohydride (LAB) reagents are a new class of powerful and selective reagents developed in the laboratory of Professor Bakthan Singaram at the University of California, Santa Cruz.
Lithium aminoborohydride (LAB) reagents are a new class of powerful and selective reagents developed in the laboratory of Professor Bakthan Singaram at the University of California, Santa Cruz.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico


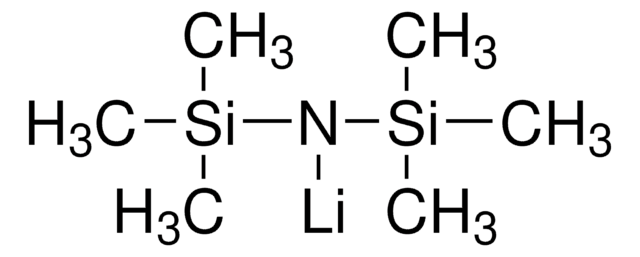
![[1,1′-bis(difenilfosfino)ferroceno]dicloropaladio(II), complejo con diclorometano](/deepweb/assets/sigmaaldrich/product/structures/825/986/4317978b-1256-4c82-ab74-6a6a3ef948b1/640/4317978b-1256-4c82-ab74-6a6a3ef948b1.png)
![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)



