374733
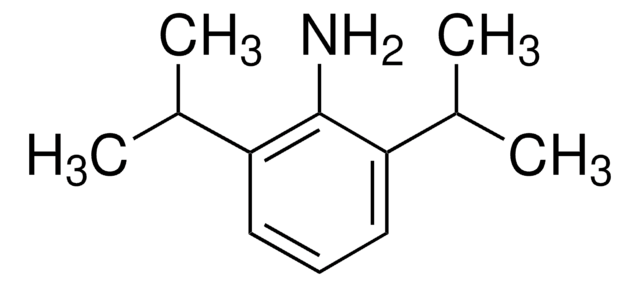
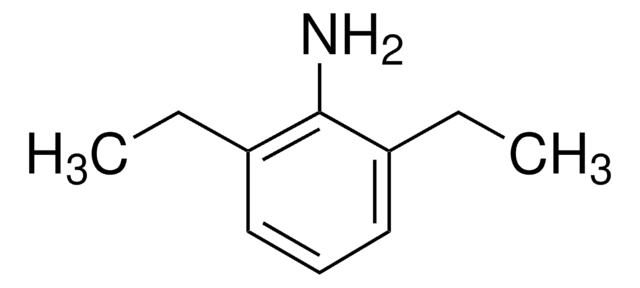
2,6-Diisopropylaniline
97%
Sinónimos:
2,6-Bis(1-methylethyl)benzenamine, 2,6-Bis(propan-2-yl)aniline, 2,6-Diisopropylphenylamine
About This Item
Productos recomendados
vapor pressure
<0.01 mmHg ( 20 °C)
Quality Level
assay
97%
form
liquid
refractive index
n20/D 1.532 (lit.)
bp
257 °C (lit.)
mp
−45 °C (lit.)
density
0.94 g/mL at 25 °C (lit.)
SMILES string
CC(C)c1cccc(C(C)C)c1N
InChI
1S/C12H19N/c1-8(2)10-6-5-7-11(9(3)4)12(10)13/h5-9H,13H2,1-4H3
InChI key
WKBALTUBRZPIPZ-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
General description
2,6-Diisopropylaniline is an important organic intermediate widely used to synthesize plastics and dyes.
2,6-Diisopropylaniline is an important organic intermediate widely used to synthesize plastics and dyes.
Application
signalword
Warning
hcodes
Hazard Classifications
Aquatic Chronic 3 - Eye Irrit. 2
Storage Class
10 - Combustible liquids
wgk_germany
WGK 2
ppe
Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Global Trade Item Number
| Número de referencia del producto (SKU) | GTIN |
|---|---|
| 374733-25G | 4061831834758 |
| 374733-5G | 4061836820367 |
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico