169978
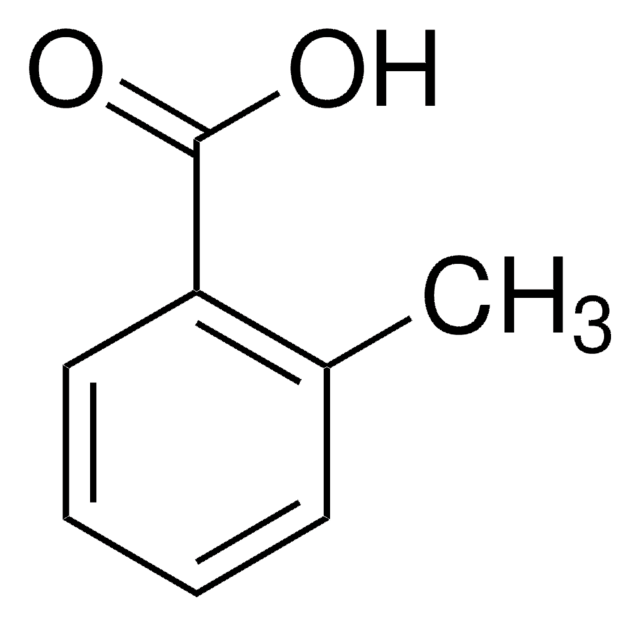
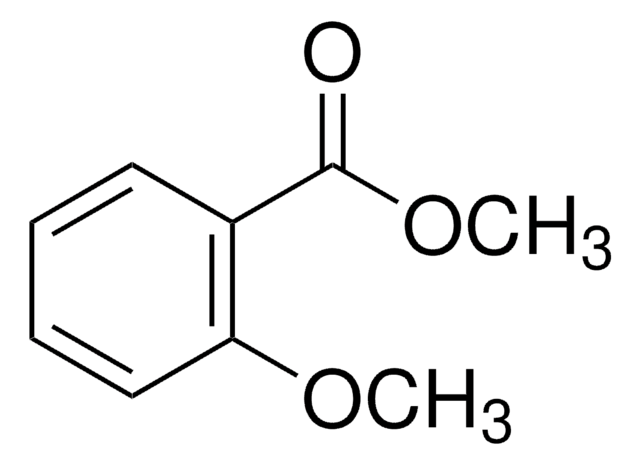
2-Methoxybenzoic acid
ReagentPlus®, 99%
Sinónimos:
O-Methylsalicylic acid, o-Anisic acid
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
CH3OC6H4CO2H
Número de CAS:
Peso molecular:
152.15
Beilstein/REAXYS Number:
509929
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
Quality Level
product line
ReagentPlus®
assay
99%
form
powder
mp
98-100 °C (lit.)
SMILES string
COc1ccccc1C(O)=O
InChI
1S/C8H8O3/c1-11-7-5-3-2-4-6(7)8(9)10/h2-5H,1H3,(H,9,10)
InChI key
ILUJQPXNXACGAN-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
2-Methoxybenzoic acid was added as carbon supplement in the culture medium of Moraxella osloensis. Photophysics of 2-methoxybenzoic acid has been investigated using both the time-correlated single photon counting and the fluorescence up-conversion techniques.
Application
2-Methoxybenzoic acid was used as internal standard during quantification of free and conjugated salicylic acid in tomato (Lycopersicon esculentum) cells by HPLC. It was also employed in the synthesis of pthalides.
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
291.2 °F - closed cup
flash_point_c
144 °C - closed cup
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Heterocycles, 39, 47-47 (1994)
Involvement of endogenous salicylic acid content, lipoxygenase and antioxidant enzyme activities in the response of tomato cell suspension cultures to NaCl.
Molina A, et al.
The New phytologist, 156(3), 409-415 (2002)
The photophysics of salicylic acid derivatives in aqueous solution.
Pozdnyakov IP, et al.
Journal of the Physical Society of Japan, 22(5), 449-454 (2009)
R L Crawford et al.
Journal of bacteriology, 121(3), 794-799 (1975-03-01)
Gentisate:oxygen 1,2-oxidoreductase (decyclizing) (EC 1.13.11.4; gentisate 1,2-dioxygenase) from Moraxella osloensis was purified to homogeneity as shown by polyacrylamide gel electrophoresis. The enzyme has a molecular weight of about 154,000 and gives rise to subunits of molecular weight 40,000 in the
T Sasaki et al.
Applied radiation and isotopes : including data, instrumentation and methods for use in agriculture, industry and medicine, 50(5), 905-909 (1999-04-24)
For in vivo measurement of the hydroxyl radical (.OH), we synthesized [11C]salicylic acid, [11C]O-acetylsalicylic acid and [11C]2-methoxybenzoic acid by carboxylation of 2-bromomagnesiumanisol using [11C]CO2. The radiochemical yield of [11C]salicylic acid, [11C]O-acetylsalicylic acid and [11C]2-methoxybenzoic acid calculated from trapped [11C]CO2 in
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico