F5062
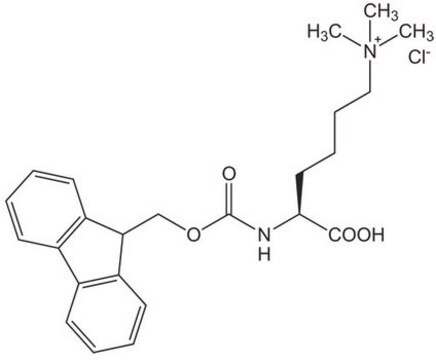
Fmoc-Lys(Me)3-OH Chloride
≥97%, for peptide synthesis
Synonym(s):
Fmoc-Lys(Me3Cl)-OH, N-alpha-Fmoc-N-alpha-trimethyl-N-L-lysine · chloride
About This Item
Recommended Products
product name
Fmoc-Lys(Me)3-OH Chloride, ≥97%
Assay
≥97%
form
liquid
reaction suitability
reaction type: Fmoc solid-phase peptide synthesis
application(s)
peptide synthesis
functional group
Fmoc
shipped in
dry ice
storage temp.
−20°C
InChI
1S/C24H30N2O4.ClH/c1-26(2,3)15-9-8-14-22(23(27)28)25-24(29)30-16-21-19-12-6-4-10-17(19)18-11-5-7-13-20(18)21;/h4-7,10-13,21-22H,8-9,14-16H2,1-3H3,(H-,25,27,28,29);1H/t22-;/m0./s1
InChI key
XUJRNPVABVHOAJ-FTBISJDPSA-N
Related Categories
General description
Application
- Synthesis of peptide linkers for monoclonal antibody-auristatin F conjugates.
- Preparation of multifunctional reagents with enhanced ionization properties for the analysis of protein modification in human cells and dynamic profiling of protein lipidation.
- Sequential peptide ligation to synthesize histone H3 containing a trimethyl lysine residue, with modified tail region.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service