857661
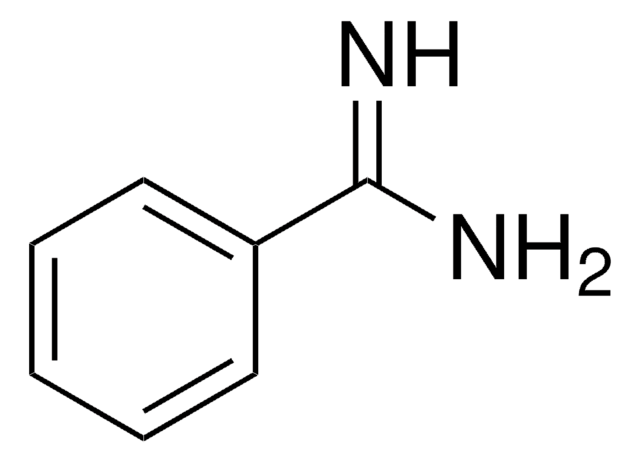
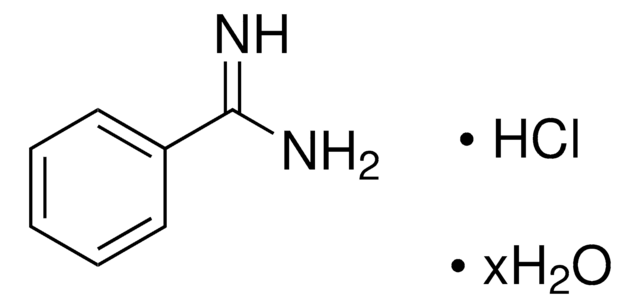
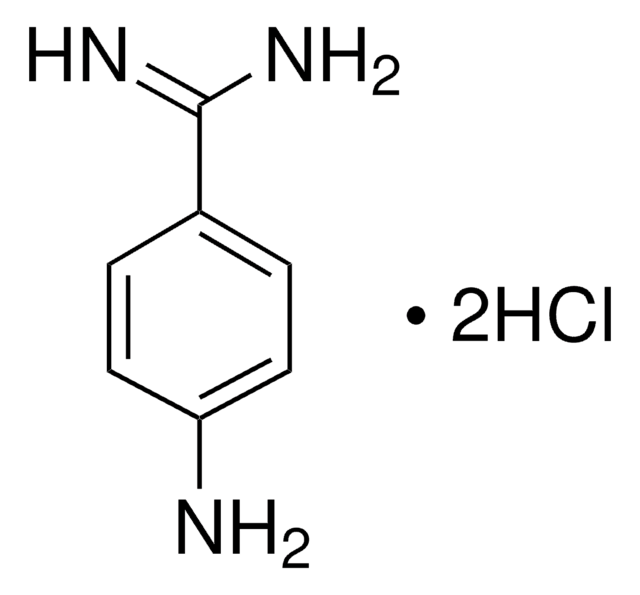
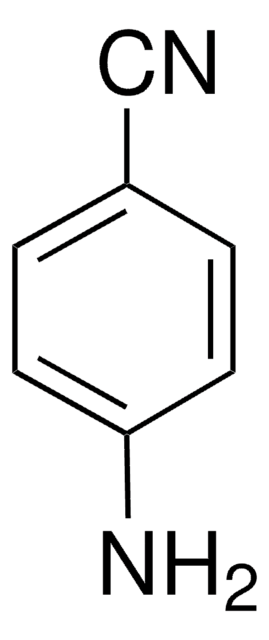
4-Aminobenzamidine dihydrochloride
98%
Synonym(s):
p-Aminobenzimidamide dihydrochloride
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
H2NC6H4C(=NH)NH2·2HCl
CAS Number:
Molecular Weight:
208.09
Beilstein:
3692927
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
crystals
mp
>300 °C (lit.)
functional group
amine
storage temp.
2-8°C
SMILES string
Cl[H].Cl[H].NC(=N)c1ccc(N)cc1
InChI
1S/C7H9N3.2ClH/c8-6-3-1-5(2-4-6)7(9)10;;/h1-4H,8H2,(H3,9,10);2*1H
InChI key
GHEHNICLPWTXJC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
4-Aminobenzamidine dihydrochloride can be used to synthesize:
- Orally active fibrinogen receptor antagonists based on benzamidines.
- Benzamidine derivatives that are selective and potent serine protease inhibitors.
- Novel pyrrolo [3,2-c] quinolines that are structural analogs of topoisomerase inhibitors such as coralyne and fagaronine.
4-Aminobenzamidine dihydrochloride is used as a ligand in affinity chromatography for purification and immobilization of enzymes.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Biochemical and molecular modeling analysis of the ability of two p-aminobenzamidine-based sorbents to selectively purify serine proteases (fibrinogenases) from snake venoms.
De-Simone S G, et al.
Journal of Chromatography. B, Analytical Technologies in the Biomedical and Life Sciences, 822(1-2), 1-9 (2005)
In vitro blood compatibility of polymeric biomaterials through covalent immobilization of an amidine derivative.
Gouzy M F, et al.
Biomaterials, 25(17), 3493-3501 (2004)
A L Nguyen et al.
Biotechnology and bioengineering, 34(9), 1186-1190 (1989-11-01)
Reactive polymers have been prepared by copolymeriz-ing N-isopropyl acrylamide (NIPAM) with N-acryloxy-succinimide (NASI) or glycidyl methacrylate (GMA). The amino groups of ligands could react with the residues of NASI or GMA and the polymers could be precipitated by temperature and/or
A L Nguyen et al.
Enzyme and microbial technology, 12(9), 663-668 (1990-09-01)
A reactive water-soluble polymer was synthesized by copolymerizing N-isopropylacrylamide and glycidyl acrylate. The reactive polymer could react with the amino groups of enzymes/proteins or other ligands to form an affinity polymer. As a model, the reactive polymer was allowed to
Specific adsorption of serine proteases on coated silica beads substituted with amidine derivatives.
S Khamlichi et al.
Journal of chromatography, 510, 123-132 (1990-06-27)
Amidine derivatives interact with serine proteases, the inhibition being due to interactions between amidine functions and the active sites of the enzymes. Five different types of amidine (substituted or unsubstituted) were coupled to coated silica beads, which had previously been
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service