418080
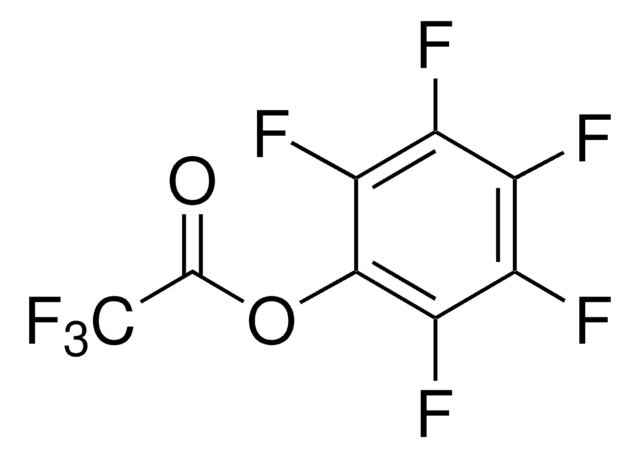
Pentafluorophenyl diphenylphosphinate
Synonym(s):
FDPP, Diphenylphosphinic acid pentafluorophenyl ester
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(C6H5)2P(O)OC6F5
CAS Number:
Molecular Weight:
384.24
MDL number:
UNSPSC Code:
12352101
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
form
solid
Quality Level
mp
47-50 °C (lit.)
functional group
fluoro
SMILES string
Fc1c(F)c(F)c(OP(=O)(c2ccccc2)c3ccccc3)c(F)c1F
InChI
1S/C18H10F5O2P/c19-13-14(20)16(22)18(17(23)15(13)21)25-26(24,11-7-3-1-4-8-11)12-9-5-2-6-10-12/h1-10H
InChI key
OOWSDKUFKGVADH-UHFFFAOYSA-N
General description
Pentafluorophenyl diphenylphosphinate (FDPP) is a reagent, used as a coupling agent in the amide bond forming reactions without racemization. It can also be employed in the synthesis of dipeptides with high yields and good optical purity. FDPP can be prepared from diphenylphosphinic chloride and pentafluorophenol in the presence of imidazole.
Application
Catalyst involved in coupling and macrocyclization
Reagent used in:
Reagent used in:
- Fmoc solid-phase synthesis
- Synthesis of phorboxazoles, furan-based cyclic homoligopeptides, and ziziphine N
FDPP can be used as a coupling reagent in the:
- Synthesis of peptides by solid-phase and solution-phase reactions.
- Preparation of macrocyclic peptide cyclotheonamide B as a thrombin inhibitor.
- Cyclooligomerization of N-methylated-L-valine thiazole amino acid to obtain its cyclic tetramers.
- Macrocyclization of an intermediate for the total synthesis of ziziphine N , and for the macrolactamization of precursor in synthesizing cryptophycin D.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Highly convergent route to cyclopeptide alkaloids. Total synthesis of ziziphine N.
He G, et al.
Organic Letters, 9(7), 1367-1369 (2007)
Synthesis of novel N-methylated thiazole-based cyclic octa-and dodecapeptides.
Dudin L, et al.
Tetrahedron, 61(5), 1257-1267 (2005)
Total synthesis of cryptophycins. Revision of the structures of cryptophycins A and C.
Barrow R A, et al.
Journal of the American Chemical Society, 117(9), 2479-2490 (1995)
Pentafluorophenyl diphenylphosphinate a new efficient coupling reagent in peptide chemistry.
Chen S and Xu J
Tetrahedron Letters, 32(46), 6711-6714 (1991)
Gang He et al.
Organic letters, 9(7), 1367-1369 (2007-03-10)
[structure: see text]. A highly convergent protocol to cyclopeptide alkaloids, as demonstrated by the first total synthesis of antiplasmodial agent ziziphine N, is developed. The key elements include construction of its aryl ether unit via Mitsunobu reaction, installation of its
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service


![Sodium tetrakis[3,5-bis(trifluoromethyl)phenyl]borate](/deepweb/assets/sigmaaldrich/product/structures/251/439/7a621e74-bfd1-4a43-833c-09adfcc1e0b3/640/7a621e74-bfd1-4a43-833c-09adfcc1e0b3.png)