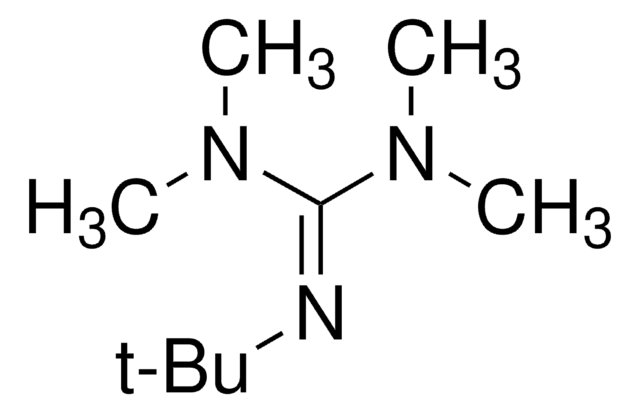
197602
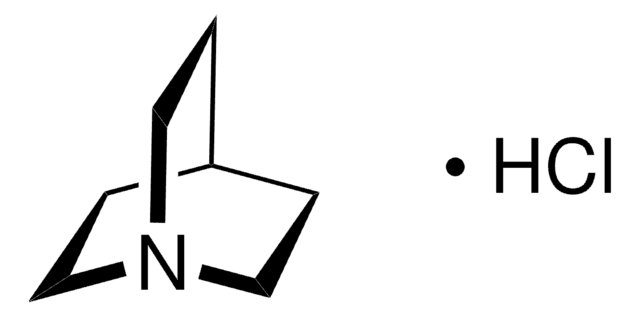
Quinuclidine
97%
Synonym(s):
1-Azabicyclo[2.2.2]octane, ABCO
About This Item
Recommended Products
vapor pressure
1.5 mmHg ( 20 °C)
Quality Level
Assay
97%
form
solid
mp
157-160 °C (lit.)
solubility
H2O: very slightly soluble
H2O: very soluble
alcohol: miscible
diethyl ether: miscible
organic solvents: very soluble
SMILES string
C1CN2CCC1CC2
InChI
1S/C7H13N/c1-4-8-5-2-7(1)3-6-8/h7H,1-6H2
InChI key
SBYHFKPVCBCYGV-UHFFFAOYSA-N
Gene Information
rat ... Chrm1(25229)
Looking for similar products? Visit Product Comparison Guide
General description
Application
- In the Baylis-Hillman reaction of aldehydes with methyl acrylate.
- For the epimerization of α-methylglucose to α-methylallose.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 2 Dermal - Acute Tox. 3 Oral - Eye Dam. 1 - Skin Irrit. 2
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Global Trade Item Number
| SKU | GTIN |
|---|---|
| 197602-10G | 4061838762528 |
| 197602-1G | 4061826732038 |
| 197602-250MG |
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service![1,4-Diazabicyclo[2.2.2]octane ReagentPlus®, ≥99%](/deepweb/assets/sigmaaldrich/product/structures/366/129/a6ff4175-974d-4fac-9038-b35e508ef252/640/a6ff4175-974d-4fac-9038-b35e508ef252.png)
![1,8-Diazabicyclo[5.4.0]undec-7-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/120/564/5b373e23-1624-489c-8efb-692de0f96ffb/640/5b373e23-1624-489c-8efb-692de0f96ffb.png)
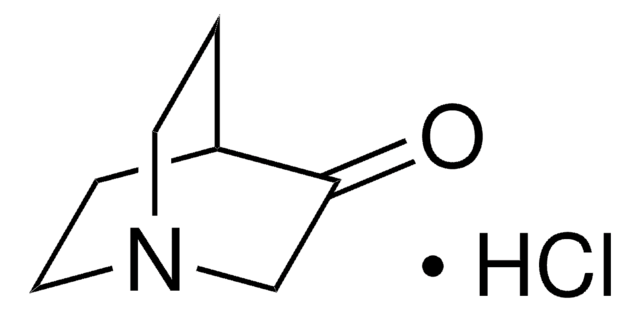
![1,5,7-Triazabicyclo[4.4.0]dec-5-ene 98%](/deepweb/assets/sigmaaldrich/product/structures/171/446/333d560c-cff6-4958-b489-5acfb3057cce/640/333d560c-cff6-4958-b489-5acfb3057cce.png)
![(Ir[dF(CF3)ppy]2(dtbpy))PF6](/deepweb/assets/sigmaaldrich/product/structures/982/913/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09/640/02dd8ddd-6deb-40a0-ab9b-07b18f1abb09.png)