221171
Pinacol
98%
Synonym(s):
2,3-Dimethyl-2,3-butanediol, Tetramethylethylene glycol
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
HOC(CH3)2C(CH3)2OH
CAS Number:
Molecular Weight:
118.17
Beilstein:
1340501
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
form
solid
bp
171-172 °C/739 mmHg (lit.)
mp
40-43 °C (lit.)
solubility
alcohol: freely soluble
diethyl ether: freely soluble
hot water: freely soluble
functional group
hydroxyl
SMILES string
CC(C)(O)C(C)(C)O
InChI
1S/C6H14O2/c1-5(2,7)6(3,4)8/h7-8H,1-4H3
InChI key
IVDFJHOHABJVEH-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
The pinacol coupling reaction with the inorganic electride [Ca2N](+)·e(−) as an electron donor in organic solvents was studied.
Application
Pinacol is a 1,2-diol that can be used:
- As a ligand to prepare uranyl complexes by reacting with uranyl nitrate and acetate hydrates.
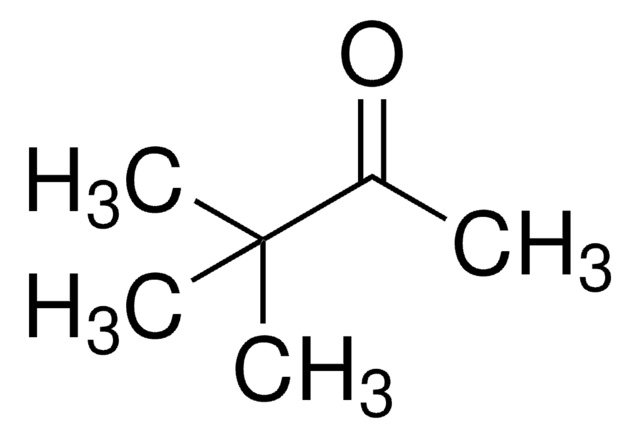
- To prepare pinacolone and 2,3-dimethyl-1,3-butadiene by typical pinacol rearrangement reaction.
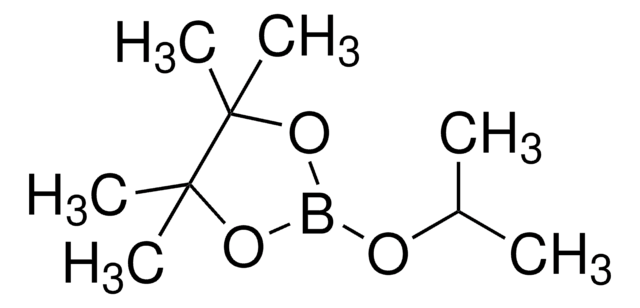
- As a reactant to synthesize 4-(4,4,5,5-tetramethyl-1,3,2-bioxaborolan-2-yl)benzoic acid, which is used to prepare derivatives of 4-acetoxy-3-phenylbenzaldehyde.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
170.6 °F - closed cup
Flash Point(C)
77 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Uranyl complexes with 1, 2-diols and tetrahydrofurfuryl alcohols
Villiers C, et al.
Polyhedron, 46(1), 133-138 (2012)
Fe-substituted molecular sieves as catalysts in liquid phase pinacol rearrangement
Hsien M, et al.
J. Mol. Catal. A: Chem., 181(1-2), 189-200 (2002)
Ye Ji Kim et al.
Chemical communications (Cambridge, England), 50(37), 4791-4794 (2014-04-02)
The scalable pinacol coupling reaction is realized utilizing the inorganic electride [Ca2N](+)·e(-) as an electron donor in organic solvents. The bond cleavages of the [Ca2N](+) layers by methanol play a vital role in transferring anionic electrons to electrophilic aldehydes, accompanying
Egor V Verbitskiy et al.
Journal of fluorescence, 25(3), 763-775 (2015-03-31)
Novel donor-π-acceptor dyes bearing the pyrimidine unit as an electron-withdrawing group have been synthesized by using combination of two processes, based on the microwave-assisted Suzuki cross-coupling reaction and nucleophilic aromatic substitution of hydrogen. Spectral properties of the obtained dyes in
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service