172050
漆树酸-CAS 16611-84-0-Calbiochem
The Anacardic Acid, also referenced under CAS 16611-84-0, modulates the biological activity of Anacardic. This small molecule/inhibitor is primarily used for Cell Structure applications.
别名:
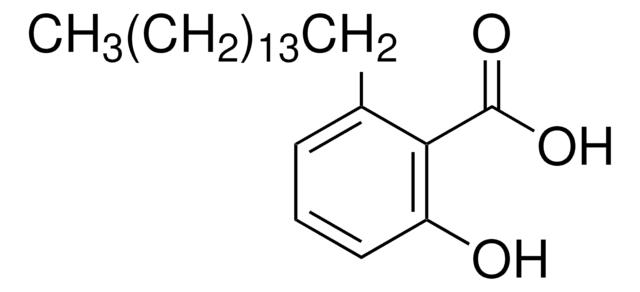
Anacardic Acid, AA, 2-Hydroxy-6-pentadecylbenzoic Acid, 6-Pentadecylsalicylic Acid, Aurora Kinase A Activator, Histone Acetyltransferase Inhibitor III, SUMOylation Inhibitor II, HAT Inhibitor III, p300/CBP Inhibitor III, PCAF Inhibitor I
登录查看公司和协议定价
所有图片(1)
About This Item
推荐产品
品質等級
化驗
≥95% (HPLC)
形狀
solid
製造商/商標名
Calbiochem®
儲存條件
OK to freeze
protect from light
顏色
white
溶解度
DMSO: 10 mg/mL
ethanol: 10 mg/mL
methanol: 10 mg/mL
運輸包裝
ambient
儲存溫度
2-8°C
InChI
1S/C22H36O3/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-16-19-17-15-18-20(23)21(19)22(24)25/h15,17-18,23H,2-14,16H2,1H3,(H,24,25)
InChI 密鑰
ADFWQBGTDJIESE-UHFFFAOYSA-N
一般說明
A cell-permeable ginkgolic acid (Cat. No. 345887) analog that inhibits protein SUMO (Cat. Nos. 662037, 662039, and 662042) modification (IC50 = 2.2 µM using RanGAP1-C2 as substrate) in an ATP-dependent manner by selectively targeting SUMO-activating enzyme E1 (Cat. Nos. 662073 and 662074) and interfering with E1-SUMO intermediate formation. Both anacardic acid and ginkgolic acid are shown to effectively decrease overall SUMOylation of 293T cellular proteins in a dose-dependent manner, while neither compound is effective in affecting overall cellular protein ubiquitination or histone H4K8 acetylation in 293T cultures, although anacardic acid is shown to inhibit p300 (Cat. No. 506200) and PCAF (Cat. No. 124026) histone acetyltransferase activities in cell-free acetylase assays (by 82% and 86%, respectively, at 10 µM). Also reported to inhibit the activity of prostaglandin synthase, tyrosinase, and lipoxygenase, as well as to enhance Aurora kinase A (Cat. No. 481413), but not Aurora kinase B, autophosphorylation and kinase activity by inducing conformation change and enhancing ATP binding.
A cell-permeable ginkgolic acid (Cat. No. 345887) analog that inhibits protein SUMO (Cat. Nos. 662037, 662039, and 662042) modification (IC50 = 2.2 µM using RanGAP1-C2 as substrate) in an ATP-dependent manner by selectively targeting SUMO-activating enzyme E1 (Cat. Nos. 662073 and 662074) and interfering with E1-SUMO intermediate formation. Both anacardic acid and ginkgolic acid are shown to effectively decrease overall SUMOylation of 293T cellular proteins in a dose-dependent manner, while neither compound is effective in affecting overall cellular protein ubiquitination or histone H4K8 acetylation in 293T cultures, although anacardic acid is shown to inhibit p300 (Cat. No. 506200) and PCAF (Cat. No. 124026) histone acetyltransferase activities in cell-free acetylase assays (by 82% and 86%, respectively, at 10 µM). Also reported to inhibit the activity of prostaglandin synthase, tyrosinase, and lipoxygenase, as well as to enhance Aurora kinase A (Cat. No. 481413), but not Aurora kinase B, autophosphorylation and kinase activity by inducing conformation change and enhancing ATP binding.
生化/生理作用
Cell permeable: yes
Primary Target
HAT
HAT
Product competes with ATP.
Reversible: no
Target IC50: 5 µM against HAT; 8.5 µM against PCAF; 2.2 µM inhibiting protein SUMO modification using RanGAP1-C2 as substrate
包裝
Packaged under inert gas
警告
Toxicity: Standard Handling (A)
重構
Following reconstitution aliquot and freeze (-20°C). Stock solutions are stable for up to 3 months at -20°C.
其他說明
Fukuda, I., et al. 2009. Chem. Biol.16, 133.
Kishore, A.H. 2008. J. Med. Chem.51, 792.
Balasubramanyam, K., et al. 2003. J. Biol. Chem.278, 19134.
Paramashivappa, R., et al. 2002. J. Agric. Food Chem.50, 7709.
Kishore, A.H. 2008. J. Med. Chem.51, 792.
Balasubramanyam, K., et al. 2003. J. Biol. Chem.278, 19134.
Paramashivappa, R., et al. 2002. J. Agric. Food Chem.50, 7709.
法律資訊
Sold under license of U.S. Patent 7,332,629 and related patents.
CALBIOCHEM is a registered trademark of Merck KGaA, Darmstadt, Germany
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门