365572
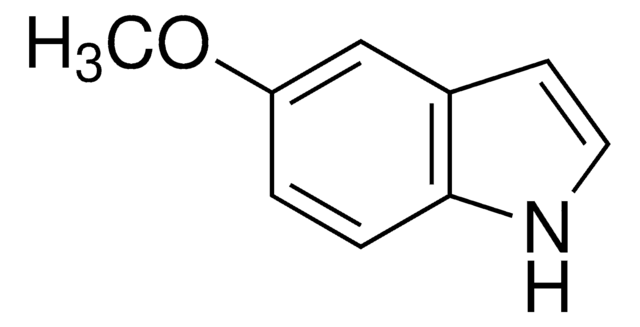
Methyl 6-methoxy-2-indolecarboxylate
99%
Synonym(s):
2-Methoxycarbonyl-6-methoxyindole, 6-Methoxyindole-2-carboxylic acid methyl ester
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C11H11NO3
CAS Number:
Molecular Weight:
205.21
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
assay
99%
mp
117-119 °C (lit.)
SMILES string
COC(=O)c1cc2ccc(OC)cc2[nH]1
InChI
1S/C11H11NO3/c1-14-8-4-3-7-5-10(11(13)15-2)12-9(7)6-8/h3-6,12H,1-2H3
InChI key
OPUUCOLVBDQWEY-UHFFFAOYSA-N
Application
Methyl 6-methoxy-2-indolecarboxylate is suitable for use in the production of dyes by Escherichia coli expressing naphthalene dioxygenase (NDO) and toluene dioxygenase (TDO). It is also suitable for use in the production of dyes by Escherichia coli expressing multicomponent phenol hydroxylase (mPH) from Pseudomonas sp. strains KL33 and KL28.
Reactant for preparation of:
- Benzoxazole containing indole analogs as peroxisome proliferator-activated receptor-γ/δ dual agonists
- Potent antiproliferative agent against human leukemia K562 cells
- Indole-indolone scaffold via [3+2] annulation of arynes
- Latonduine derivatives via intramolecular Heck reaction as possible anticancer agents
- Arylthioindoles as potent inhibitors of tubulin polymerization
- Heterocycle-fused derivatives of 1-oxo-1,2,3,4-tetrahydropyrazine via Ugi condensation
- Indole fatty alcohols (IFAs) as promoters of differentiation of neural stem cell derived neurospheres into neurons. Potential application for treatment of neurodegenerative diseases
- Light-dependent tumor necrosis factor-α antagonists
- 2-substituted indole melatonin receptor ligands
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
J Y Kim et al.
Letters in applied microbiology, 36(6), 343-348 (2003-05-20)
To isolate and characterize the phorate [O,O-diethyl-S-(ethylthio)methyl phosphoradiothioate] degrading bacteria from agricultural soil, and their assessment for multifarious biological activities of environmental and agronomic significance. Based on their morphological and biochemical characteristics, the selected isolates PS-1, PS-2 and PS-3 were
J Y Kim et al.
Letters in applied microbiology, 41(2), 163-168 (2005-07-22)
To establish multicomponent phenol hydroxylases (mPHs) as novel biocatalysts for producing dyestuffs and hydroxyindoles such as 7-hydroxyindole (7-HI) from indole and its derivatives. We have isolated Pseudomonas sp. KL33, which possesses a phenol degradation pathway similar to that found in
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service