Y0001256
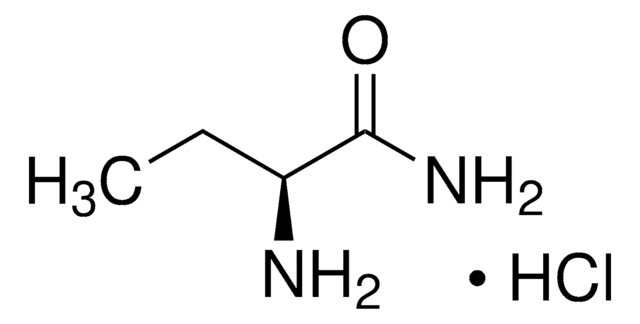
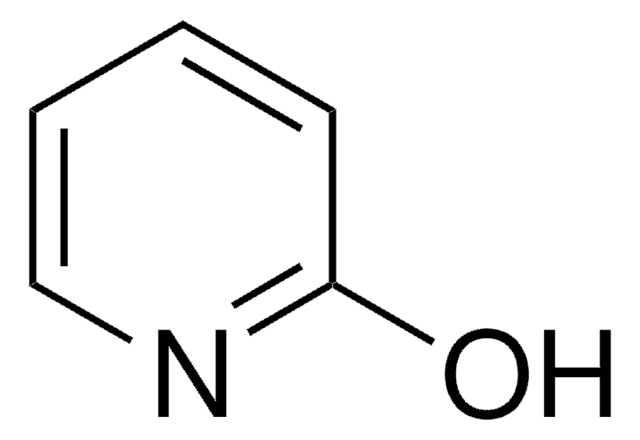
Levetiracetam impurity C
European Pharmacopoeia (EP) Reference Standard
Synonim(y):
2-Hydroxypyridine, 2(1H)-Pyridone, 2-Pyridinol
About This Item
Polecane produkty
klasa czystości
pharmaceutical primary standard
rodzina API
levetiracetam
producent / nazwa handlowa
EDQM
tw
280-281 °C (lit.)
mp
105-107 °C (lit.)
Zastosowanie
pharmaceutical (small molecule)
format
neat
temp. przechowywania
2-8°C
ciąg SMILES
Oc1ccccn1
InChI
1S/C5H5NO/c7-5-3-1-2-4-6-5/h1-4H,(H,6,7)
Klucz InChI
UBQKCCHYAOITMY-UHFFFAOYSA-N
Szukasz podobnych produktów? Odwiedź Przewodnik dotyczący porównywania produktów
Opis ogólny
Zastosowanie
Opakowanie
Inne uwagi
Hasło ostrzegawcze
Danger
Zwroty wskazujące rodzaj zagrożenia
Zwroty wskazujące środki ostrożności
Klasyfikacja zagrożeń
Acute Tox. 3 Oral
Kod klasy składowania
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Klasa zagrożenia wodnego (WGK)
WGK 3
Temperatura zapłonu (°F)
Not applicable
Temperatura zapłonu (°C)
Not applicable
Choose from one of the most recent versions:
Certyfikaty analizy (CoA)
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact Obsługa Klienta
Masz już ten produkt?
Dokumenty związane z niedawno zakupionymi produktami zostały zamieszczone w Bibliotece dokumentów.
Klienci oglądali również te produkty
Nasz zespół naukowców ma doświadczenie we wszystkich obszarach badań, w tym w naukach przyrodniczych, materiałoznawstwie, syntezie chemicznej, chromatografii, analityce i wielu innych dziedzinach.
Skontaktuj się z zespołem ds. pomocy technicznej