377406
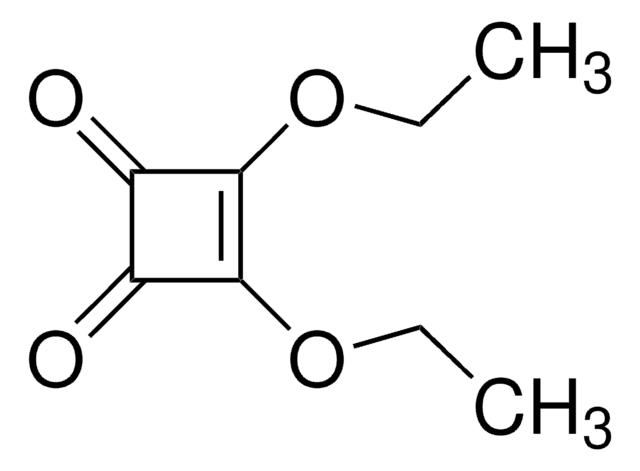
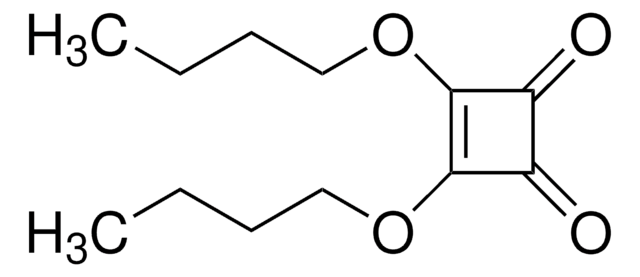
3,4-Dimethoxy-3-cyclobutene-1,2-dione
99%
Synonym(s):
Dimethyl squarate
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(CH3O)2C4(=O)2
CAS Number:
Molecular Weight:
142.11
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
form
solid
mp
55-57 °C (lit.)
storage temp.
2-8°C
SMILES string
COC1=C(OC)C(=O)C1=O
InChI
1S/C6H6O4/c1-9-5-3(7)4(8)6(5)10-2/h1-2H3
InChI key
SZBNZTGCAMLMJY-UHFFFAOYSA-N
Related Categories
General description
3,4-Dimethoxy-3-cyclobutene-1,2-dione (Dimethyl squarate) is a cyclobutene derivative. It reacts with hydroxylamine derivatives to afford 3-hydroxyamino-4-methoxy-3-cyclobutene-1,2-diones.
Application
3,4-Dimethoxy-3-cyclobutene-1,2-dione (Dimethyl squarate) may be used in the synthesis of the following compounds:
- chiral squaramides, highly enantioselective catalyst for the Friedel-Crafts reactions of indoles
- 3-(hydroxyamino)-4-methoxy-3-cyclobutene-1,2-dione
- squarate derivatives of the O-SP-core antigens
- methyl squarate derivative of the Ogawa O-SP-core antigen
- o-quinodimethanes
- benzocyclobutenes
- quinones
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Resp. Sens. 1 - Skin Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Peng Xu et al.
Bioconjugate chemistry, 22(10), 2179-2185 (2011-09-09)
Bacterial O-SP-core antigens can be conjugated to proteins in the same, simple way as synthetic, linker-equipped carbohydrates by applying squaric acid chemistry. Introduction of spacers (linkers) to either O-SP-core antigens or protein carriers, which is involved in commonly applied protocols
Kerriann K Badal et al.
Cell reports, 26(3), 507-517 (2019-01-17)
Mechanisms that regulate the bi-directional transport of mitochondria in neurons for maintaining functional synaptic connections are poorly understood. Here, we show that in the pre-synaptic sensory neurons of the Aplysia gill withdrawal reflex, the formation of functional synapses leads to
Hydroxylamin-Derivate der Quadratsaure.
Zinner G, et al.
Arch. Pharm. (Weinheim), 318(11), 977-983 (1985)
The Journal of Organic Chemistry, 57, 6896-6896 (1992)
Tetrahedron Letters, 34, 6177-6177 (1993)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service