MAB3392
Anti-Collagen Type III Antibody, clone IE7-D7
clone 1E7-D7, from mouse
Sinónimos:
collagen, type III, alpha 1, collagen, fetal, Ehlers-Danlos syndrome type IV, autosomal dominant, alpha1 (III) collagen, collagen alpha-1(III) chain
About This Item
Productos recomendados
biological source
mouse
Quality Level
antibody form
purified immunoglobulin
antibody product type
primary antibodies
clone
1E7-D7, monoclonal
species reactivity
rat
species reactivity (predicted by homology)
human (based on 100% sequence homology)
technique(s)
ELISA: suitable
immunohistochemistry: suitable
western blot: suitable
isotype
IgG1κ
NCBI accession no.
UniProt accession no.
shipped in
wet ice
target post-translational modification
unmodified
Gene Information
human ... COL3A1(1281)
General description
Specificity
Immunogen
Application
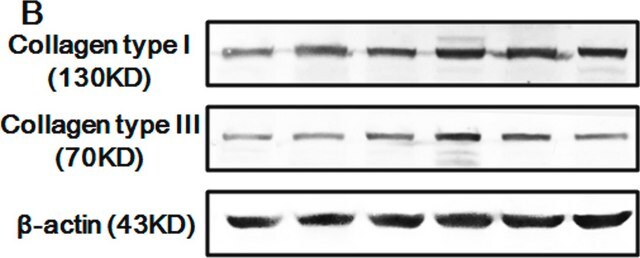
Western Blot Analysis: A previous lot of this antibody was used to detect collagen type III in western blot under non-reduced conditions (Werkmeister J.A., et al., 1988; Ramshaw, J.S., et al., 1988).
Some Collagen samples can be contaminated with other Collagen Types. When purified Collagen is used in an application the purity of the Collagen sample should be verified by SDS-page to minimize the risk of false positives.
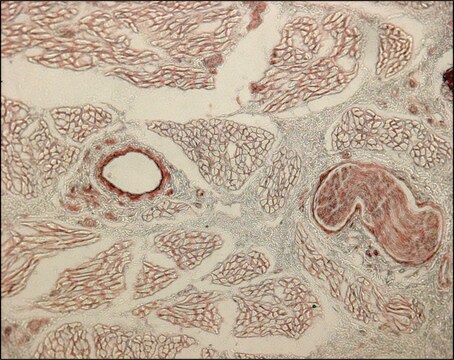
Immunohistochemistry Analysis: A previous lot of this antibody was used to detect collagen type III in immunohistochemistry (Werkmeister J.A., et al., 1989; Werkmeister J.A., et al., 1989; Werkmeister J.A., et al., 1988).
Cell Structure
ECM Proteins
Quality
Immunohistochemistry Analysis: A 1:600 dilution of this antibody detected Collagen Type III in rat knee joint tissue.
Target description
Physical form
Storage and Stability
Analysis Note
Rat knee joint tissue
Other Notes
Disclaimer
¿No encuentra el producto adecuado?
Pruebe nuestro Herramienta de selección de productos.
Storage Class
12 - Non Combustible Liquids
wgk_germany
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico