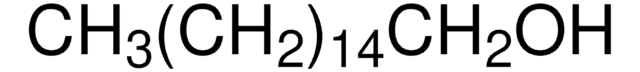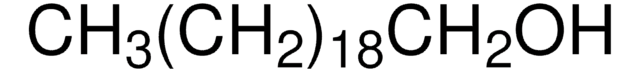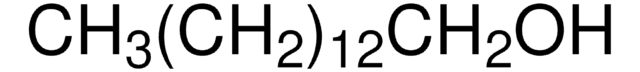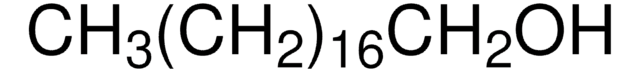추천 제품
Grade
certified reference material
pharmaceutical secondary standard
Quality Level
Agency
traceable to USP 1367699
CofA
current certificate can be downloaded
포장
pkg of 5X50 mg
응용 분야
pharmaceutical
InChI
1S/C18H32O/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19/h3-4,6-7,9-10,19H,2,5,8,11-18H2,1H3/b4-3-,7-6-,10-9-
InChI key
IKYKEVDKGZYRMQ-PDBXOOCHSA-N
유사한 제품을 찾으십니까? 방문 제품 비교 안내
일반 설명
Pharmaceutical secondary standards for application in quality control provide pharma laboratories and manufacturers with a convenient and cost-effective alternative to the preparation of in-house working standards
분석 메모
These secondary standards offer multi-traceability to the USP, EP and BP primary standards, where they are available.
기타 정보
This Certified Reference Material (CRM) is produced and certified in accordance with ISO 17034 and ISO/IEC 17025. All information regarding the use of this CRM can be found on the certificate of analysis.
각주
To see an example of a Certificate of Analysis for this material enter LRAA6959(changes by product) in the Documents slot below. This is an example certificate only and may not be the lot that you receive.
Storage Class Code
10 - Combustible liquids
WGK
awg
Flash Point (°F)
Not applicable
Flash Point (°C)
Not applicable
Choose from one of the most recent versions:
시험 성적서(COA)
Lot/Batch Number
Sorry, we don't have COAs for this product available online at this time.
If you need assistance, please contact 고객 지원 부서
자사의 과학자팀은 생명 과학, 재료 과학, 화학 합성, 크로마토그래피, 분석 및 기타 많은 영역을 포함한 모든 과학 분야에 경험이 있습니다..
고객지원팀으로 연락바랍니다.