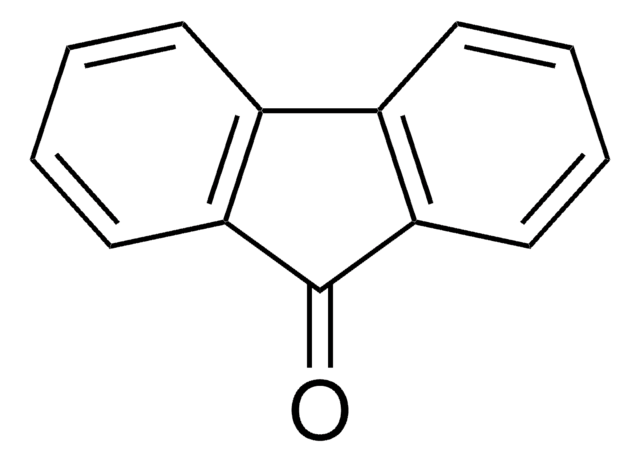
P10801
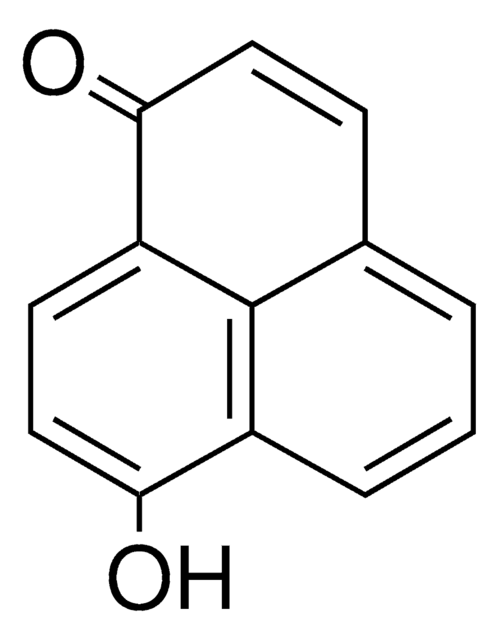
Perinaphthenone
97%
Synonym(s):
1H-Benzonaphthen-1-one, 7-Perinaphthenone, Phenalenone, Phenalone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C13H8O
CAS Number:
Molecular Weight:
180.20
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
powder
mp
153-156 °C (lit.)
SMILES string
O=C1C=Cc2cccc3cccc1c23
InChI
1S/C13H8O/c14-12-8-7-10-4-1-3-9-5-2-6-11(12)13(9)10/h1-8H
InChI key
WWBGWPHHLRSTFI-UHFFFAOYSA-N
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Mahmoud Fahmi Elsebai et al.
Organic & biomolecular chemistry, 9(3), 802-808 (2010-11-26)
The marine-derived fungus Coniothyrium cereale was isolated from the green alga Enteromorpha sp. and found to produce the new phenalenone derivatives 1-7 as well as the known compounds lactone 8, (-) sclerodin (9), lamellicolic anhydride (10), (-) scleroderolide (11), and
Christopher M Whidbey et al.
Water research, 46(16), 5287-5296 (2012-08-11)
Steroid estrogens are endocrine disrupting contaminants frequently detected in natural waters. Because these estrogens can elicit significant biological responses in aquatic organisms, it is important to study their rates and pathways of degradation in natural waters and to identify whether
Zhiyong Guo et al.
Magnetic resonance in chemistry : MRC, 45(5), 439-441 (2007-03-21)
The complete (1)H and (13)C NMR assignments are reported for the novel natural product Bacillosporin D together with the known compound Bacillosporin C. These compounds containing seven rings were isolated from the mangrove endophytic fungus SBE-14 from the South China
Cristina Flors et al.
Accounts of chemical research, 39(5), 293-300 (2006-05-17)
Plants defend themselves from pathogen infections or mechanical injury by a number of mechanisms, including the induced biosynthesis of antimicrobial secondary metabolites. These compounds, termed phytoalexins, represent a very economical way to counteract hazard, because the carbon and energy resources
William Hidalgo et al.
Journal of agricultural and food chemistry, 57(16), 7417-7421 (2009-07-28)
The levels of native fungitoxic perinaphthenone phytoalexins in susceptible Musa varieties (banana), which are commercially grown in large plantations, are too low to provide plants with long-lasting protection against highly pathogenic fungi. Novel strategies for plant protection are necessary to
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service
![Cyclopenta[d,e,f]phenanthrene 97%](/deepweb/assets/sigmaaldrich/product/structures/107/640/eed40ce0-e715-4438-9cb3-dc3dc13dcb9b/640/eed40ce0-e715-4438-9cb3-dc3dc13dcb9b.png)