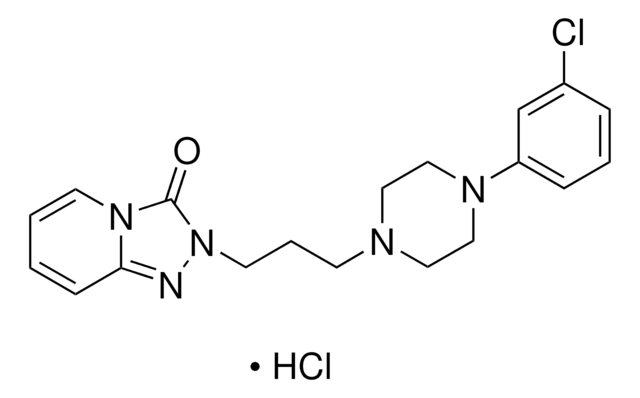
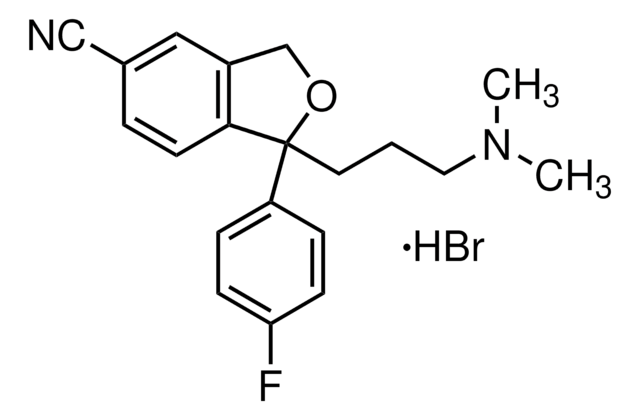
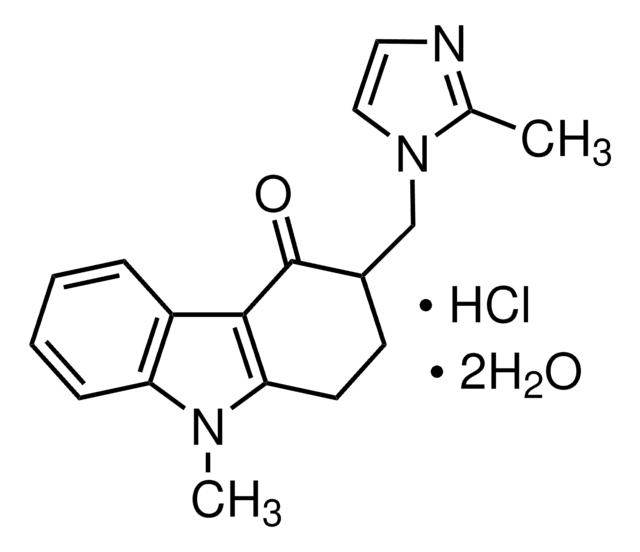
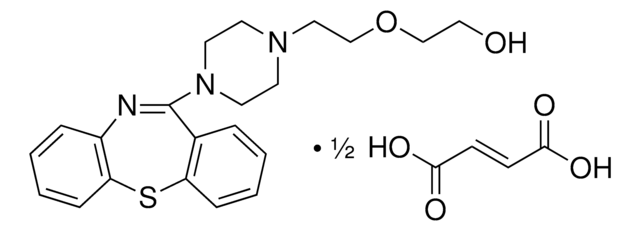
おすすめの製品
グレード
certified reference material
pharmaceutical secondary standard
品質水準
認証
traceable to BP 1137
traceable to Ph. Eur. R1000600
traceable to USP 1604654
APIファミリー
risperidone
CofA
current certificate can be downloaded
包装
pkg of 500 mg
テクニック
HPLC: suitable
gas chromatography (GC): suitable
アプリケーション
pharmaceutical (small molecule)
フォーマット
neat
保管温度
2-30°C
SMILES記法
CC1=C(CCN2CCC(CC2)c3noc4cc(F)ccc34)C(=O)N5CCCCC5=N1
InChI
1S/C23H27FN4O2/c1-15-18(23(29)28-10-3-2-4-21(28)25-15)9-13-27-11-7-16(8-12-27)22-19-6-5-17(24)14-20(19)30-26-22/h5-6,14,16H,2-4,7-13H2,1H3
InChI Key
RAPZEAPATHNIPO-UHFFFAOYSA-N
遺伝子情報
human ... DRD2(1813) , HTR2A(3356) , HTR2C(3358)
類似した製品をお探しですか? 訪問 製品比較ガイド
詳細
アプリケーション
アナリシスノート
その他情報
脚注
おすすめ製品
関連製品
シグナルワード
Danger
危険有害性情報
注意書き
危険有害性の分類
Acute Tox. 3 Oral
保管分類コード
6.1C - Combustible, acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
WGK
WGK 3
引火点(°F)
Not applicable
引火点(℃)
Not applicable
Choose from one of the most recent versions:
試験成績書(COA)
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
この製品を見ている人はこちらもチェック
ライフサイエンス、有機合成、材料科学、クロマトグラフィー、分析など、あらゆる分野の研究に経験のあるメンバーがおります。.
製品に関するお問い合わせはこちら(テクニカルサービス)