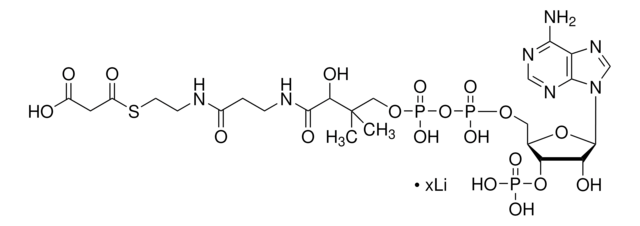
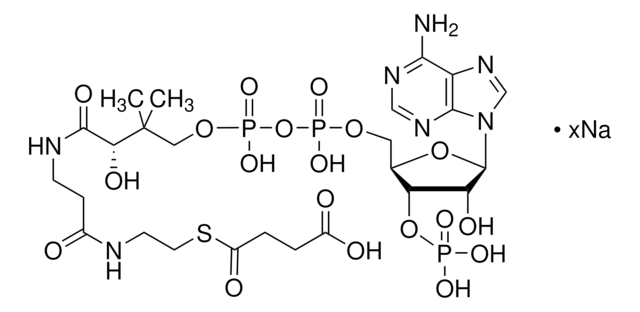
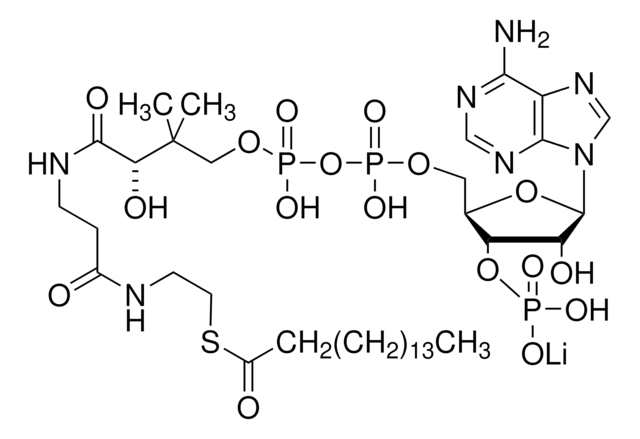
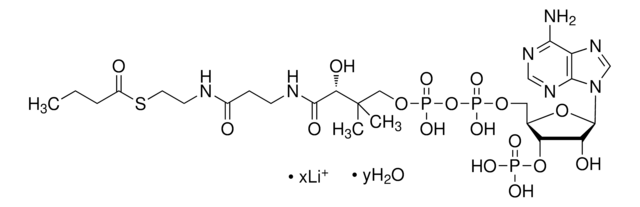
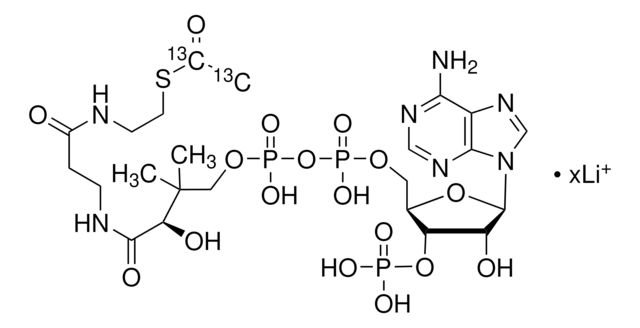
A2056
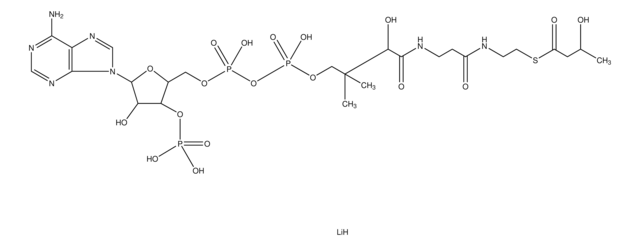
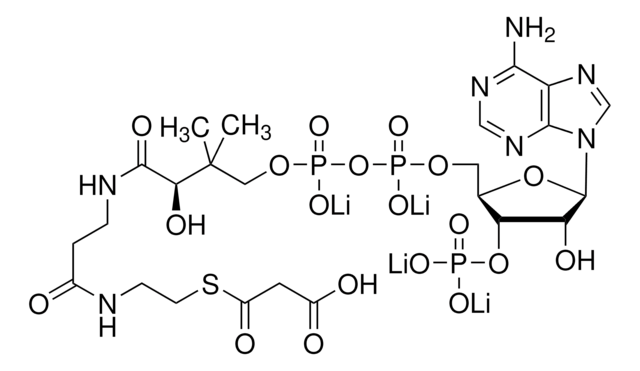
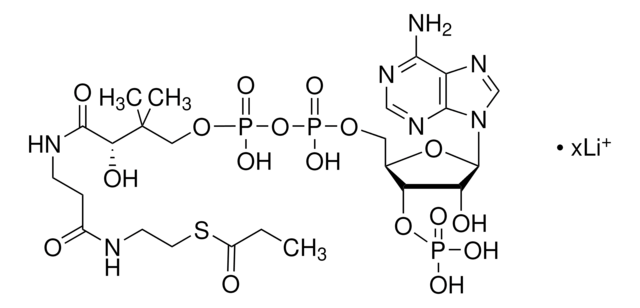
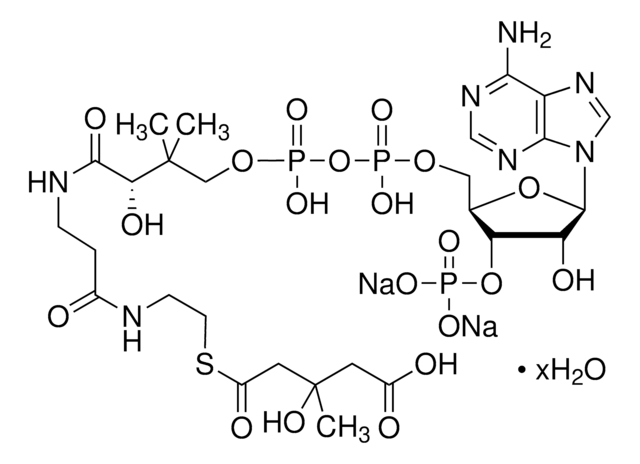
Acetyl coenzyme A trisodium salt
≥93% (HPLC), powder
Synonym(s):
Acetyl-S- CoA, Acetyl CoA
About This Item
Recommended Products
Quality Level
Assay
≥93% (HPLC)
form
powder
solubility
H2O: 100 mg/mL
storage temp.
−20°C
SMILES string
O[C@H]1[C@](O[C@@H]([C@H]1OP(O)(O)=O)COP(OP(OCC(C)(C)[C@@H](O)C(NCCC(NCCSC(C)=O)=O)=O)(O)=O)(O)=O)([H])N2C3=NC=NC(N)=C3N=C2.[3Na]
InChI
1S/C23H38N7O17P3S.Na/c1-12(31)51-7-6-25-14(32)4-5-26-21(35)18(34)23(2,3)9-44-50(41,42)47-49(39,40)43-8-13-17(46-48(36,37)38)16(33)22(45-13)30-11-29-15-19(24)27-10-28-20(15)30;/h10-11,13,16-18,22,33-34H,4-9H2,1-3H3,(H,25,32)(H,26,35)(H,39,40)(H,41,42)(H2,24,27,28)(H2,36,37,38);/q;+1/p-1/t13-,16-,17-,18?,22-;/m1./s1
InChI key
HNLIOWFIXSPFEC-WLYMNMRISA-M
Gene Information
human ... CHAT(1103) , HAT1(8520) , KAT2A(2648) , KAT2B(8850) , KAT5(10524)
mouse ... HAT1(107435) , KAT2A(14534) , KAT2B(18519) , KAT5(81601)
rat ... HAT1(296501) , KAT2A(303539) , KAT2B(301164) , KAT5(192218)
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
Biochem/physiol Actions
Preparation Note
Other Notes
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Regulatory Listings
Regulatory Listings are mainly provided for chemical products. Only limited information can be provided here for non-chemical products. No entry means none of the components are listed. It is the user’s obligation to ensure the safe and legal use of the product.
JAN Code
A2056-10MG:4548174006108
A2056-1MG:4548174006115
A2056-PM:
A2056-BULK:
A2056-5MG:4548174006139
A2056-25MG:4548174006122
A2056-VAR:
A2056-100MG:4548174006092
A2056-10MG-KC:
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Sigma-Aldrich presents an article about how proliferatively active cells require both a source of carbon and of nitrogen for the synthesis of macromolecules. Although a large proportion of tumor cells utilize aerobic glycolysis and shunt metabolites away from mitochondrial oxidative phosphorylation, many tumor cells exhibit increased mitochondrial activity.
Information on fatty acid synthesis and metabolism in cancer cells. Learn how proliferatively active cells require fatty acids for functions such as membrane generation, protein modification, and bioenergetic requirements. These fatty acids are derived either from dietary sources or are synthesized by the cell.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service