745723
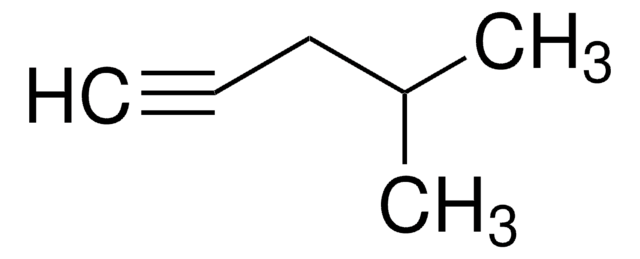
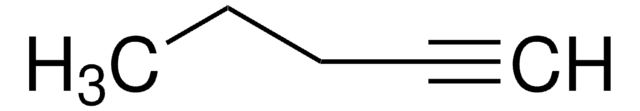
3-Methyl-1-butyne
95%
Sinonimo/i:
1-Isopentyne, 2-Methyl-3-butyne, 3,3-Dimethyl-1-propyne, Isopropylacetylene, Isopropylethyne
Autenticatiper visualizzare i prezzi riservati alla tua organizzazione & contrattuali
About This Item
Formula empirica (notazione di Hill):
C5H8
Numero CAS:
Peso molecolare:
68.12
Numero CE:
Numero MDL:
Codice UNSPSC:
12352100
ID PubChem:
NACRES:
NA.22
Prodotti consigliati
Saggio
95%
Forma fisica
liquid
Indice di rifrazione
n20/D 1.374
Densità
0.666 g/mL at 25 °C (lit.)
Temperatura di conservazione
2-8°C
Stringa SMILE
C#CC(C)C
InChI
1S/C5H8/c1-4-5(2)3/h1,5H,2-3H3
USCSRAJGJYMJFZ-UHFFFAOYSA-N
Applicazioni
3-Methyl-1-butyne can be used as a reactant in the ruthenium-catalyzed synthesis of functionalized cycloheptadienes and titanium-catalyzed [2+2+1] multicomponent pyrrole synthesis. It is also used as a precursor in the total syntheses of (+)-frondosin A, (−)-citrinadin A, (+)-citrinadin B and coraxeniolide A.
3-Methyl-1-butyne is a terminal alkyne that can be used to prepare:
- Maleimide-fused cyclopentenones by reacting with N-substituted maleimides via Co2(CO)8-mediated Pauson−Khand reaction.
- An alkyne carboxylic acid (Alkyne Segment C) intermediate, which is used in the total synthesis of oscillatoxin D and its analogs.
Certificati d'analisi (COA)
Cerca il Certificati d'analisi (COA) digitando il numero di lotto/batch corrispondente. I numeri di lotto o di batch sono stampati sull'etichetta dei prodotti dopo la parola ‘Lotto’ o ‘Batch’.
Possiedi già questo prodotto?
I documenti relativi ai prodotti acquistati recentemente sono disponibili nell’Archivio dei documenti.
I clienti hanno visto anche
Enantioselective total synthesis of (-)-citrinadin A and revision of its stereochemical structure.
Bian, Zhiguo and Marvin, Christopher C and Martin, Stephen F
Journal of the American Chemical Society, 135(30), 10886-10889 (2013)
Synthetic Route to Oscillatoxin D and Its Analogues
Nokura Y, et al.
Organic Letters, 19(21), 5992-5995 (2017)
Total synthesis of (+)-frondosin A. Application of the Ru-catalyzed [5+ 2] cycloaddition.
Trost, Barry M and Hu, Yimin and Horne, Daniel B
Journal of the American Chemical Society, 129(38), 11781-11790 (2007)
Metal carbene-promoted sequential transformations for the enantioselective synthesis of highly functionalized cycloheptadienes.
Deng L, et al.
Journal of the American Chemical Society, 127(5), 1342-1343 (2005)
An unconventional approach to the enantioselective synthesis of caryophylloids.
Larionov, Oleg V and Corey, EJ
Journal of the American Chemical Society, 130(10), 2954-2955 (2008)
Il team dei nostri ricercatori vanta grande esperienza in tutte le aree della ricerca quali Life Science, scienza dei materiali, sintesi chimica, cromatografia, discipline analitiche, ecc..
Contatta l'Assistenza Tecnica.