Chiral Phosphoric Acids: Versatile Organocatalysts with Expanding Applications
Introduction
Advantages
Representative Applications
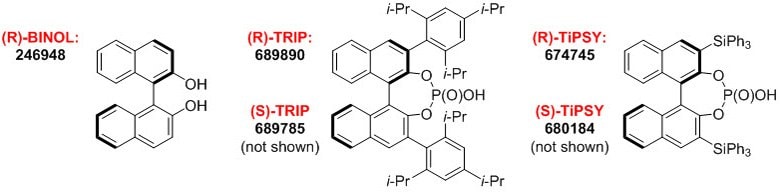
Product Information
Introduction
We are proud to offer a suite of BINOL-derived chiral phosphoric acids in our collection of products for asymmetric catalysis. This well established class of chiral Brønsted acid catalysts has been applied to an ever-increasing number of useful transformations.
Advantages
- Alternative to metal catalysts and chiral auxiliaries
- Both enantiomers of catalyst are available
- Relatively low catalyst loading (often 1-5 mol %)
- High selectivity at non-cryogenic reaction temperatures (-30 to 23 °C)
Representative Applications
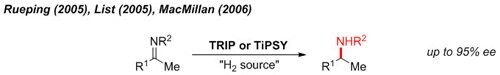
Reductive Amination
One of the earliest demonstrations of chiral phosphoric acid catalysis is the metal-free reduction of imines with an organic reductant (Hantzsch ester) to give enantioenriched amines.1
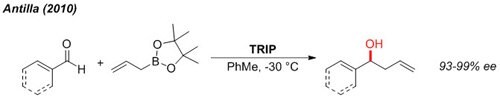
Allylation
The enantioselective allylation of aldehydes can be accomplished under very mild conditions at non-cryogenic temperature (-30 °C).2
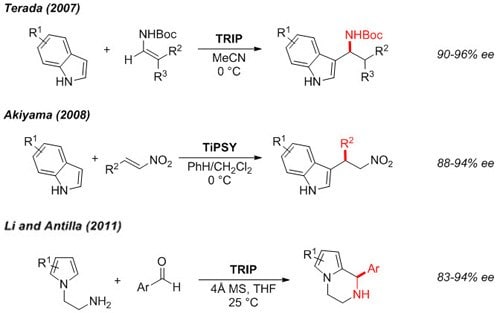
Friedel-Crafts Alkylation
Functionalized indoles3,4 and pyrroles5 can be accessed in enantioenriched form by asymmetric alkylation.
References
To continue reading please sign in or create an account.
Don't Have An Account?