09658
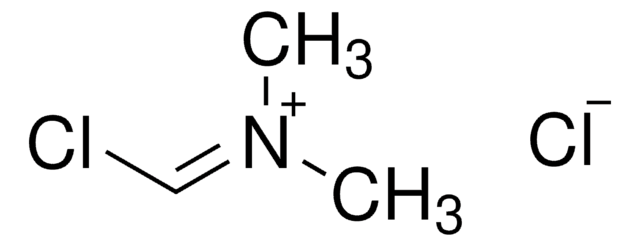
Chloro-N,N,N′,N′-tetramethylformamidinium hexafluorophosphate
≥98.0% (T)
Synonym(s):
TCFH
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C5H12ClF6N2P
CAS Number:
Molecular Weight:
280.58
Beilstein:
7896715
MDL number:
UNSPSC Code:
12352101
PubChem Substance ID:
NACRES:
NA.22
Quality Level
Assay
≥98.0% (T)
reaction suitability
reaction type: Coupling Reactions
mp
99-118 °C
application(s)
peptide synthesis
storage temp.
2-8°C
SMILES string
F[P-](F)(F)(F)(F)F.CN(C)\C(Cl)=[N+](\C)C
InChI
1S/C5H12ClN2.F6P/c1-7(2)5(6)8(3)4;1-7(2,3,4,5)6/h1-4H3;/q+1;-1
InChI key
CUKNPSDEURGZCO-UHFFFAOYSA-N
Application
Chloro-N,N,N′,N′-tetramethylformamidinium hexafluorophosphate can be used as a reactant for the synthesis of:
It can also be used as a reagent for the synthesis of:
- Onium salts for use in peptide coupling.
- Benzotriazole based uranium reagent, a safer replacement for coupling reagents.
It can also be used as a reagent for the synthesis of:
- Cancer cell cytotoxins.
- Bioconjugation reagents.
Other Notes
Coupling reagent for peptide synthesis and starting material for preparing other coupling reagents
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Certificates of Analysis (COA)
Lot/Batch Number
Don't see the Right Version?
If you require a particular version, you can look up a specific certificate by the Lot or Batch number.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
An Efficient Second-Generation Manufacturing Process for the pan-RAF Inhibitor Belvarafenib
Zell, D., et al.
Organic Process Research & Development, 25, 10, 2338-2350 (2021)
Ayman El-Faham, et al
European Journal of Organic Chemistry, 19, 3641-3649 (2010)
Judit Tulla-Puche et al.
Bioconjugate chemistry, 19(10), 1968-1971 (2008-09-23)
Prodrugs are increasingly used as delivery vehicles for pharmaceutical agents that present solubility and/or pharmacokinetic/metabolic issues. In the course of the development of prodrugs for the antitumoral agent thiocoraline, standard coupling reagents and procedures failed to provide the desired target
N, N, N′, N′-Tetramethylchloroformamidinium hexafluorophosphate (TCFH), a powerful coupling reagent for bioconjugation
Tulla-Puche J, et al.
Bioconjugate Chemistry, 19(10), 1968-1971 (2008)
J. Phillip Kennedy and Craig W. Lindsley
Tetrahedron Letters, 51, 2493-2496 (2010)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service