92549
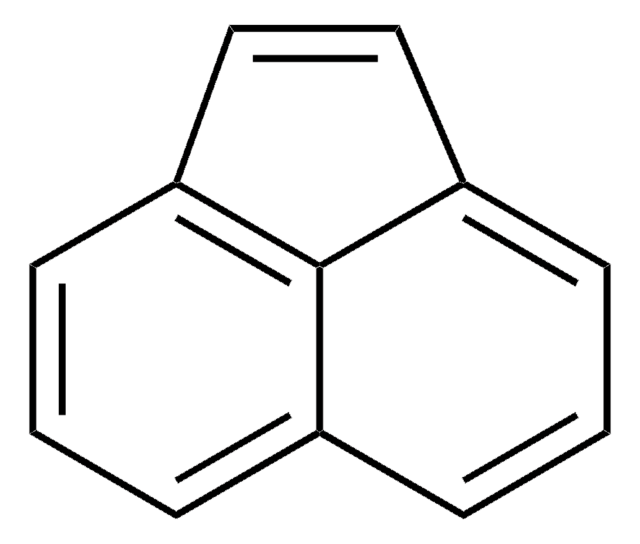
Acenaphthylene
certified reference material, TraceCERT®, Manufactured by: Sigma-Aldrich Production GmbH, Switzerland
About This Item
Productos recomendados
grado
certified reference material
TraceCERT®
Nivel de calidad
Línea del producto
TraceCERT®
caducidad
limited shelf life, expiry date on the label
fabricante / nombre comercial
Manufactured by: Sigma-Aldrich Production GmbH, Switzerland
técnicas
HPLC: suitable
gas chromatography (GC): suitable
bp
280 °C (lit.)
mp
78-82 °C (lit.)
89-92 °C
densidad
0.899 g/mL at 25 °C (lit.)
aplicaciones
environmental
formato
neat
temp. de almacenamiento
2-8°C
cadena SMILES
c1cc2C=Cc3cccc(c1)c23
InChI
1S/C12H8/c1-3-9-4-2-6-11-8-7-10(5-1)12(9)11/h1-8H
Clave InChI
HXGDTGSAIMULJN-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
Certified content by quantitative NMR incl. uncertainty and expiry date are given on the certificate.
Download your certificate at: http://www.sigma-aldrich.com.
Aplicación
Envase
Información legal
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 4 Oral - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
251.6 °F - closed cup
Punto de inflamabilidad (°C)
122.0 °C - closed cup
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Protocolos
US EPA Method 8270 (PAH only): GC Analysis of PAHs on SLB®-5ms
US EPA Method 610 describes the analysis of polynuclear aromatic hydrocarbons (commonly referred to as PAHs or PNAs) by both HPLC and GC.
GC Analysis of Polynuclear Aromatic Hydrocarbons (PAHs) in Salmon on SPB®-608 (20 m x 0.18 mm I.D., 0.18 µm) after QuEChERS Cleanup using Supel™ QuE Z-Sep, Fast GC Analysis
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico