739898
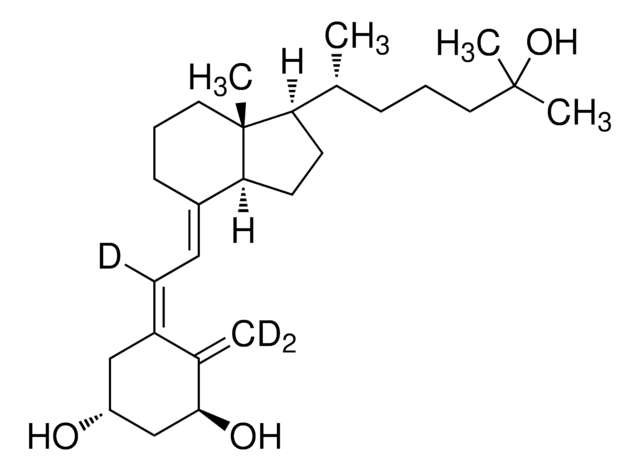
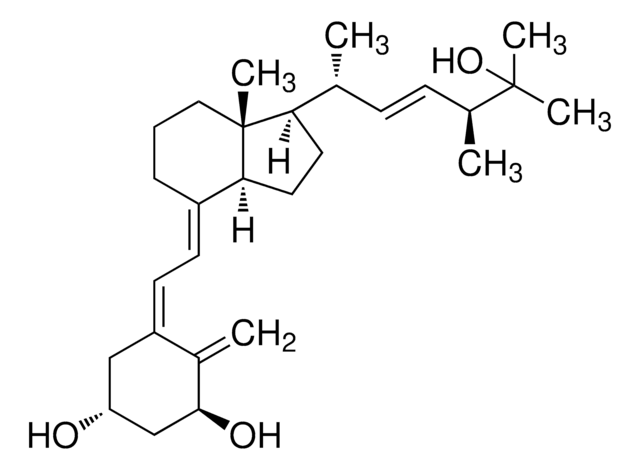
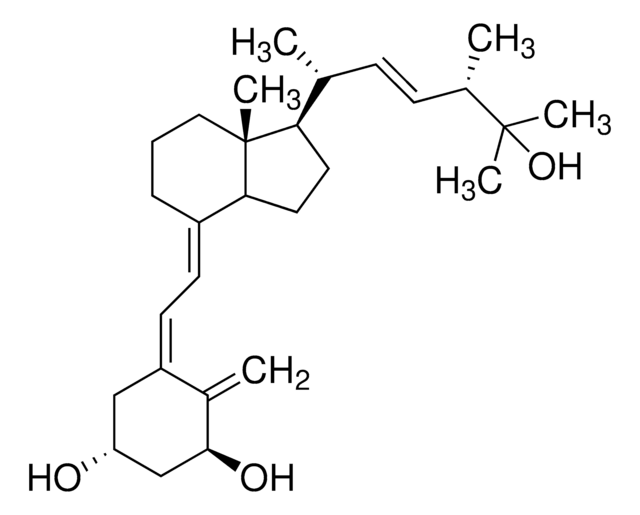
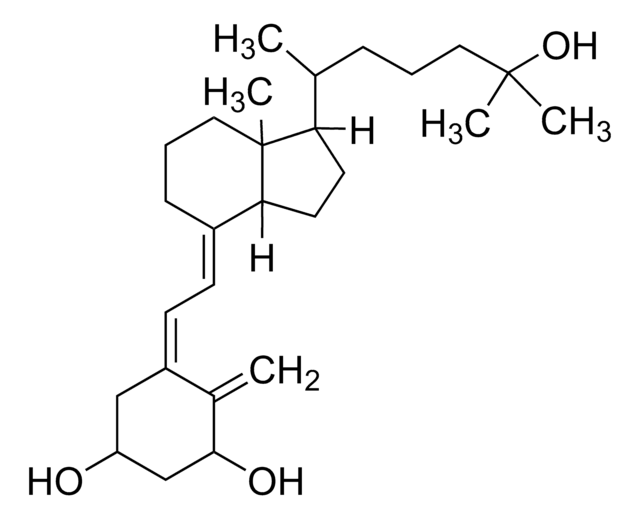
1α,25-Dihydroxyvitamin D2 solution
50 μg/mL in ethanol, 95% (CP)
Synonym(s):
1α,25-Dihydroxycalciferol solution
About This Item
Recommended Products
Quality Level
Assay
95% (CP)
form
solution
concentration
50 μg/mL in ethanol
technique(s)
mass spectrometry (MS): suitable
color
colorless
application(s)
detection
storage temp.
−20°C
SMILES string
CC(O)(C)[C@@H](C)/C=C/[C@@H](C)[C@H]1CCC([C@]1(C)CCC/2)C2=C\C=C(C[C@@H](O)C[C@@H]3O)/C3=C
InChI
1S/C28H44O3/c1-18(9-10-19(2)27(4,5)31)24-13-14-25-21(8-7-15-28(24,25)6)11-12-22-16-23(29)17-26(30)20(22)3/h9-12,18-19,23-26,29-31H,3,7-8,13-17H2,1-2,4-6H3/b10-9+,21-11+,22-12-/t18-,19+,23-,24-,25?,26+,28-/m1/s1
InChI key
ZGLHBRQAEXKACO-KPKHAADHSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Application
Packaging
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Flam. Liq. 2
Storage Class Code
3 - Flammable liquids
WGK
WGK 1
Flash Point(F)
57.2 °F - closed cup
Flash Point(C)
14 °C - closed cup
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
The metabolic role and health implications of the various vitamin D isoforms are of current clinical interest. Therefore, it is important to have an analytical method that will resolve all of the known isoforms with necessary sensitivity and specificity
This application shows an Ascentis Express F5 provided rapid resolution of the isobaric vitamin D metabolites. MS detection provides the necessary secondary resolution.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service