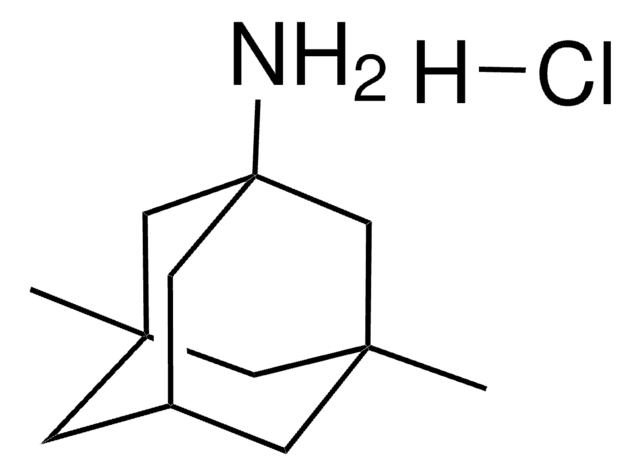
All Photos(3)
About This Item
Empirical Formula (Hill Notation):
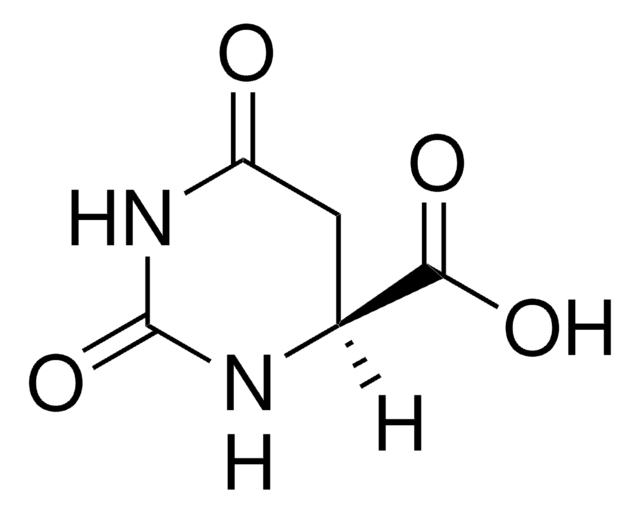
C5H6N2O4
CAS Number:
Molecular Weight:
158.11
MDL number:
UNSPSC Code:
12352204
PubChem Substance ID:
Recommended Products
form
powder
SMILES string
OC(=O)C1CC(=O)NC(=O)N1
InChI
1S/C5H6N2O4/c8-3-1-2(4(9)10)6-5(11)7-3/h2H,1H2,(H,9,10)(H2,6,7,8,11)
InChI key
UFIVEPVSAGBUSI-UHFFFAOYSA-N
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Timo Heikkilä et al.
Journal of medicinal chemistry, 50(2), 186-191 (2007-01-19)
Pyrimidine biosynthesis presents an attractive drug target in malaria parasites due to the absence of a pyrimidine salvage pathway. A set of compounds designed to inhibit the Plasmodium falciparum pyrimidine biosynthetic enzyme dihydroorotate dehydrogenase (PfDHODH) was synthesized. PfDHODH-specific inhibitors with
Joana Costeira-Paulo et al.
Cell chemical biology, 25(3), 309-317 (2018-01-24)
The interactions between proteins and biological membranes are important for drug development, but remain notoriously refractory to structural investigation. We combine non-denaturing mass spectrometry (MS) with molecular dynamics (MD) simulations to unravel the connections among co-factor, lipid, and inhibitor binding
Ceyhun Bereketoglu et al.
Physiological genomics, 49(10), 549-566 (2017-09-10)
Bioaccumulative environmental estrogen, nonylphenol (NP; 4-nonylphenol), is widely used as a nonionic surfactant and can affect human health. Since genomes of
Daniel Ken Inaoka et al.
Biochemistry, 47(41), 10881-10891 (2008-09-24)
Dihydroorotate dehydrogenase (DHOD) from Trypanosoma cruzi (TcDHOD) is a member of family 1A DHOD that catalyzes the oxidation of dihydroorotate to orotate (first half-reaction) and then the reduction of fumarate to succinate (second half-reaction) in the de novo pyrimidine biosynthesis
Genetic diversity and kinetic properties of Trypanosoma cruzi dihydroorotate dehydrogenase isoforms.
Idalia Sariego et al.
Parasitology international, 55(1), 11-16 (2005-09-21)
Dihydroorotate dehydrogenase (DHOD) is the fourth enzyme in the de novo pyrimidine biosynthetic pathway and is essential in Trypanosoma cruzi, the parasitic protist causing Chagas' disease. T. cruzi and human DHOD have different biochemical properties, including the electron acceptor capacities
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service