901606
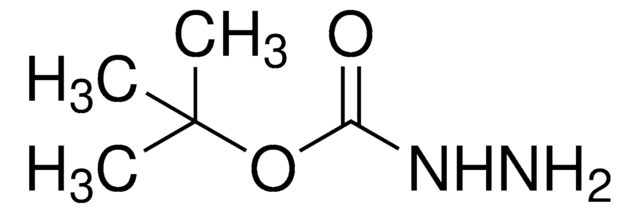
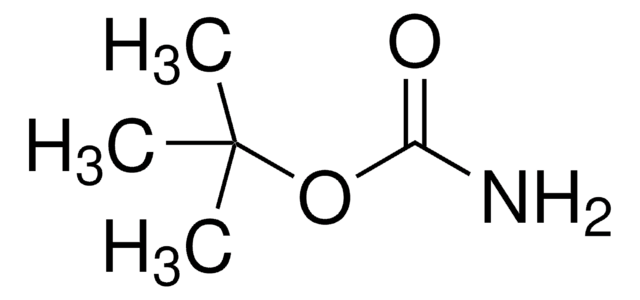
tert-Butyl carbazate solution
1.0 M in dichloromethane
Synonyme(s) :
tert-Butoxycarbonyl hydrazide, tert-Butyl hydrazinecarboxylate, Boc-hydrazide
About This Item
Produits recommandés
Forme
liquid
Concentration
1.0 M in dichloromethane
Indice de réfraction
n/D 1.427
Densité
1.290 g/mL
InChI
1S/C5H12N2O2/c1-5(2,3)9-4(8)7-6/h6H2,1-3H3,(H,7,8)
Clé InChI
DKACXUFSLUYRFU-UHFFFAOYSA-N
Application
Reagent used in solid phase peptide synthesis and in α-amino aldehyde optical purity determinations. Condenses with aldehydes to form hydrazones which are intermediates in the synthesis of HIV-1 protease inhibitors.
Produit(s) apparenté(s)
Mention d'avertissement
Warning
Mentions de danger
Conseils de prudence
Classification des risques
Carc. 2 - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Organes cibles
Central nervous system
Code de la classe de stockage
6.1C - Combustible acute toxic Cat.3 / toxic compounds or compounds which causing chronic effects
Classe de danger pour l'eau (WGK)
WGK 3
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique