E9906
Anti-EDEM2 (N-terminal) antibody produced in rabbit
~1.0 mg/mL, affinity isolated antibody, buffered aqueous solution
Synonyme(s) :
Anti-ER degradation enhancer, mannosidase alpha-like 2
About This Item
Produits recommandés
Source biologique
rabbit
Conjugué
unconjugated
Forme d'anticorps
affinity isolated antibody
Type de produit anticorps
primary antibodies
Clone
polyclonal
Forme
buffered aqueous solution
Poids mol.
antigen ~70 kDa
Espèces réactives
human, rat (predicted), mouse
Concentration
~1.0 mg/mL
Technique(s)
indirect immunofluorescence: 5-10 μg/mL using mouse 3T3 cells
western blot: 0.5-1 μg/mL using whole extracts of HEK-293T cells expressing recombinant human EDEM2
Numéro d'accès UniProt
Conditions d'expédition
dry ice
Température de stockage
−20°C
Modification post-traductionnelle de la cible
unmodified
Informations sur le gène
human ... EDEM2(55741)
mouse ... Edem2(108687)
rat ... Edem2(296304)
Description générale
Immunogène
Application
Actions biochimiques/physiologiques
Forme physique
Clause de non-responsabilité
Vous ne trouvez pas le bon produit ?
Essayez notre Outil de sélection de produits.
Code de la classe de stockage
10 - Combustible liquids
Classe de danger pour l'eau (WGK)
nwg
Point d'éclair (°F)
Not applicable
Point d'éclair (°C)
Not applicable
Équipement de protection individuelle
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
Certificats d'analyse (COA)
Recherchez un Certificats d'analyse (COA) en saisissant le numéro de lot du produit. Les numéros de lot figurent sur l'étiquette du produit après les mots "Lot" ou "Batch".
Déjà en possession de ce produit ?
Retrouvez la documentation relative aux produits que vous avez récemment achetés dans la Bibliothèque de documents.
Notre équipe de scientifiques dispose d'une expérience dans tous les secteurs de la recherche, notamment en sciences de la vie, science des matériaux, synthèse chimique, chromatographie, analyse et dans de nombreux autres domaines..
Contacter notre Service technique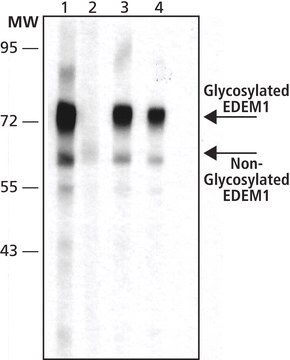
![(±)-(E)-4-Ethyl-2-[(E)-hydroxyimino]-5-nitro-3-hexenamide ≥98%](/deepweb/assets/sigmaaldrich/product/structures/404/859/0c6eb80d-2c3c-4bf1-98e8-1b319b597b12/640/0c6eb80d-2c3c-4bf1-98e8-1b319b597b12.png)