116130
Diethyl vinylphosphonate
97%
Sinônimo(s):
Vinylphosphonic acid diethyl ester
About This Item
Produtos recomendados
Ensaio
97%
índice de refração
n20/D 1.429 (lit.)
pb
202 °C (lit.)
densidade
1.068 g/mL at 25 °C (lit.)
temperatura de armazenamento
2-8°C
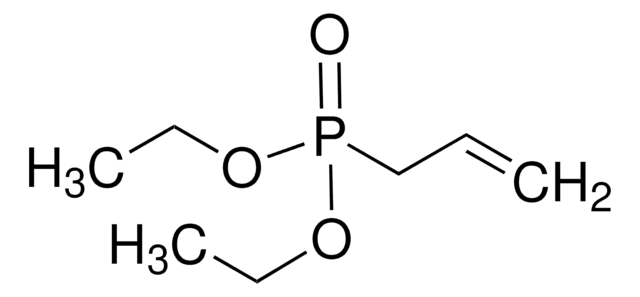
cadeia de caracteres SMILES
CCOP(=O)(OCC)C=C
InChI
1S/C6H13O3P/c1-4-8-10(7,6-3)9-5-2/h6H,3-5H2,1-2H3
chave InChI
DREPONDJUKIQLX-UHFFFAOYSA-N
Procurando produtos similares? Visita Guia de comparação de produtos
Categorias relacionadas
Aplicação
- α, β-unsaturated phosphonates by reacting with arylboronic acids via Pd-catalyzed Mizoroki−Heck reaction.
- 2-(arylamino)ethyl phosphonates by condensing with primary and secondary amines via the aza-Michael addition reaction.
It can be also employed as a monomer unit for the preparation of high-molecular-weight polymer, poly(diethyl vinylphosphonate) using lanthanide complexes.{18)
Código de classe de armazenamento
10 - Combustible liquids
Classe de risco de água (WGK)
WGK 3
Ponto de fulgor (°F)
230.0 °F - closed cup
Ponto de fulgor (°C)
110 °C - closed cup
Equipamento de proteção individual
Eyeshields, Gloves, multi-purpose combination respirator cartridge (US)
Certificados de análise (COA)
Busque Certificados de análise (COA) digitando o Número do Lote do produto. Os números de lote e remessa podem ser encontrados no rótulo de um produto após a palavra “Lot” ou “Batch”.
Já possui este produto?
Encontre a documentação dos produtos que você adquiriu recentemente na biblioteca de documentos.
Nossa equipe de cientistas tem experiência em todas as áreas de pesquisa, incluindo Life Sciences, ciência de materiais, síntese química, cromatografia, química analítica e muitas outras.
Entre em contato com a assistência técnica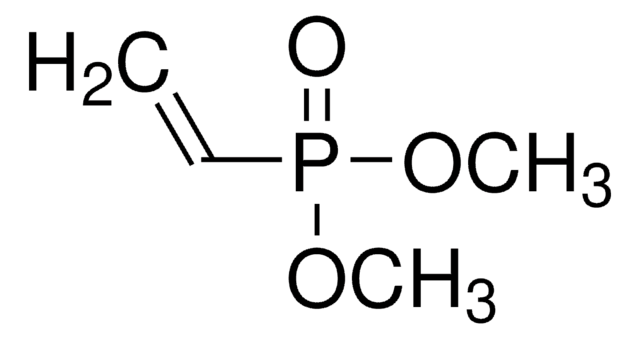


![Bis[2-(methacryloyloxy)ethyl] phosphate](/deepweb/assets/sigmaaldrich/product/structures/128/336/4e7a3e38-338c-423e-95b8-70d9d1f8e121/640/4e7a3e38-338c-423e-95b8-70d9d1f8e121.png)