D-011
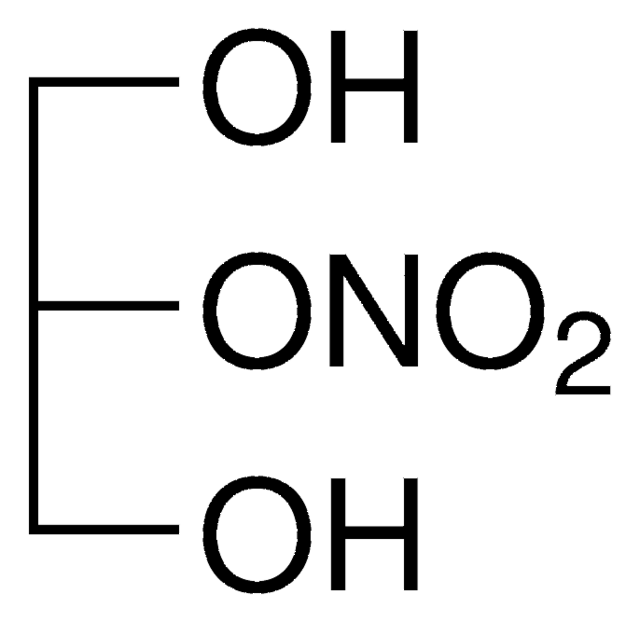
1,3-Dinitroglycerin solution
1.0 mg/mL in acetonitrile, ampule of 1 mL, certified reference material, Cerilliant®
About This Item
Recommended Products
grade
certified reference material
Quality Level
form
liquid
feature
Snap-N-Spike®/Snap-N-Shoot®
packaging
ampule of 1 mL
manufacturer/tradename
Cerilliant®
concentration
1.0 mg/mL in acetonitrile
technique(s)
gas chromatography (GC): suitable
liquid chromatography (LC): suitable
application(s)
pharmaceutical (small molecule)
format
single component solution
storage temp.
−20°C
SMILES string
OC(CO[N+]([O-])=O)CO[N+]([O-])=O
InChI
1S/C3H6N2O7/c6-3(1-11-4(7)8)2-12-5(9)10/h3,6H,1-2H2
InChI key
ASIGVDLTBLZXNC-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
- Population pharmacokinetics of nitroglycerin and of its two metabolites after a single 24-hour application of a nitroglycerin transdermal matrix delivery system: This study presents a comprehensive analysis on the population pharmacokinetics of nitroglycerin, focusing on its behavior and the behavior of its metabolites following the use of a transdermal delivery system. The findings contribute to understanding the systemic availability and action of nitroglycerin from such systems, which could be relevant for 1,3-Dinitroglycerin given its structural and functional similarities (Auclair et al., 1998).
- Novel pharmacokinetic modelling of transdermal nitroglycerin: This research explores advanced pharmacokinetic models that describe the absorption and action of nitroglycerin when administered through transdermal patches. Such models are crucial for developing effective transdermal delivery systems for similar compounds like 1,3-Dinitroglycerin (Auclair et al., 1998).
- Bioequivalence Comparison of Two Drug-in-Adhesive Transdermal Nitroglycerin Patches: This study compares the bioequivalence of two transdermal patches delivering nitroglycerin, highlighting the critical factors in the design and performance of transdermal systems that could similarly apply to 1,3-Dinitroglycerin (Harrison et al., 1996).
- Percutaneous absorption of 1,3-dinitroglycerin and a trial of pharmacokinetic analysis: Directly relevant to 1,3-Dinitroglycerin, this study examines its percutaneous absorption characteristics, offering insights into its potential applications in transdermal delivery systems (Ogiso et al., 1990).
- Analysis of solutions containing glutathione and inorganic nitrite: application to nitroglycerin metabolism studies: While primarily focused on nitroglycerin, this analysis could provide secondary insights into the metabolic pathways and interactions of 1,3-Dinitroglycerin with biomolecules like glutathione, which is crucial for understanding its pharmacological effects (Curry et al., 1987).
Legal Information
related product
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Dermal - Acute Tox. 4 Inhalation - Acute Tox. 4 Oral - Eye Irrit. 2 - Flam. Liq. 2
Storage Class Code
3 - Flammable liquids
WGK
WGK 2
Flash Point(F)
35.6 °F - closed cup
Flash Point(C)
2 °C - closed cup
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service