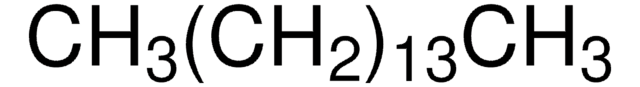410497
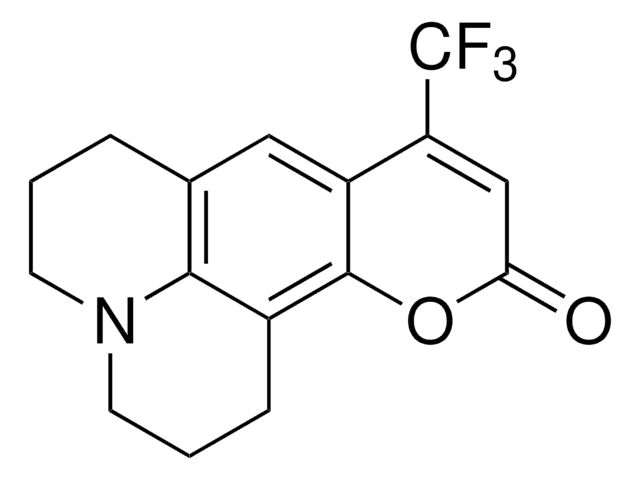
4-(Dicyanomethylene)-2-methyl-6-(4-dimethylaminostyryl)-4H-pyran
Dye content 98 %
Sinónimos:
DCM
About This Item
Productos recomendados
form
solid
Quality Level
composition
Dye content, 98%
mp
215-220 °C (lit.)
λmax
468 nm
OLED device performance
ITO/Alq3:DCM/Alq3/Mg:Ag
ITO/TPD/Alq3:DCM (10%)/Alq3/Mg:Ag
SMILES string
CN(C)c1ccc(\C=C\C2=CC(\C=C(C)O2)=C(\C#N)C#N)cc1
InChI
1S/C19H17N3O/c1-14-10-16(17(12-20)13-21)11-19(23-14)9-6-15-4-7-18(8-5-15)22(2)3/h4-11H,1-3H3/b9-6+
InChI key
YLYPIBBGWLKELC-RMKNXTFCSA-N
Categorías relacionadas
General description
Application
Features and Benefits
signalword
Danger
Hazard Classifications
Acute Tox. 4 Inhalation - Eye Irrit. 2 - Flam. Sol. 1 - Skin Irrit. 2 - STOT SE 3
target_organs
Respiratory system
Storage Class
4.1B - Flammable solid hazardous materials
wgk_germany
WGK 3
flash_point_f
109.4 °F - closed cup
flash_point_c
43 °C - closed cup
ppe
Eyeshields, Gloves, type P3 (EN 143) respirator cartridges
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Developed in the last several years, fluorescence quenching microscopy (FQM) has enabled rapid, inexpensive, and high-fidelity visualization of two-dimensional (2D) materials such as graphene-based sheets and MoS2.
Graphene has emerged as the new wonder material. Being only one atom thick and composed of carbon atoms arranged in a hexagonal honeycomb lattice structure, the interest in this material has exploded exponentially since 2004 when it was first isolated and identified using a very simple method.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico